Indicated for treatment of bleeding episodes and perioperative management in adults and children with hemophilia A or B with inhibitors, congenital factor VII (FVII) deficiency, Glanzmann’s thrombasthenia with refractoriness to platelet transfusions, and in adults with acquired hemophilia.
Financial programs and support for patients
Novo Nordisk believes providing financial support through NovoSecure™, a network that helps your eligible patients access co-pay savings, product assistance, and more.
Financial programs and support for patients
Novo Nordisk provides assistance through NovoCare®, a network to help your patients access co-pay savings, product support, and more.

NovoCare® is designed for your patients
Patients can access:
· Product support programs that help with treatment costs
· Representatives who speak Spanish to better serve them
You can help your patients by calling 1-844-668-6732 or have them contact NovoCare® at NovoCare.com.
NovoCare® is designed for your patients
Patients can access:
· Product support programs that help with treatment costs
· Representatives who speak Spanish to better serve them
You can help your patients by calling 1-844-668-6732 or have them contact NovoCare® at NovoCare.com.
NovoCare® can help with:
QuickCheck™ benefits verification
Verify your patients’ benefits and receive confirmation of coverage. If no coverage is available, names of alternative therapies may be supplied.
Patient Assistance Program (PAP)

See if your patients are eligible to receive their treatment free of charge. No registration or monthly fees required.
Eligibility and restrictions apply
Patient Trial
Program

Find out if your patients qualify to receive a limited supply of Novo Nordisk’s product for free.
Eligibility and restrictions apply
Patient
enrollment form

Download and complete the patient enrollment form to attest your patient’s need for treatment.
Insurance coordination
Help with benefits verification
We can help you verify your patient’s benefits with QuickCheck™. Submit the form to receive a Summary of Benefits, generally within 4 hours,a from 9:00 am to 4:00 pm Eastern time.
- Download the QuickCheck™ Form
- Fax it to NovoCare® at 1-866-488-6576
- Or email to: novocarespecialist@novocare.com
aQuickCheck™ benefits verification can be completed within 4 hours only if all required information has been submitted.
Additional resources for patients
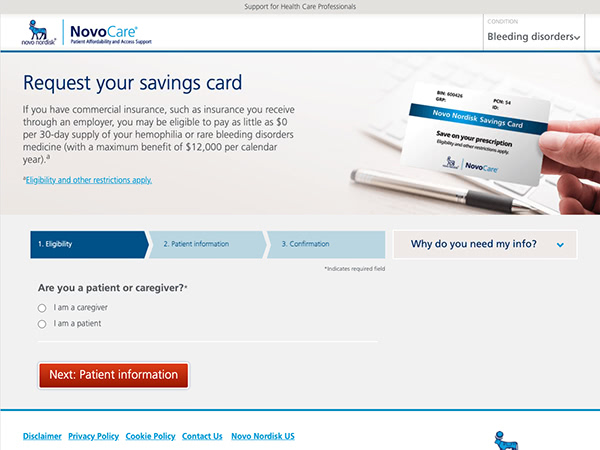
Eligibility and restrictions apply
Interim Product Program
This limited program provides therapy to qualifying patientsc during a coverage gap. Contact a NovoCare® Specialist by calling 1-844-668-6732.
Free Trial Prescription
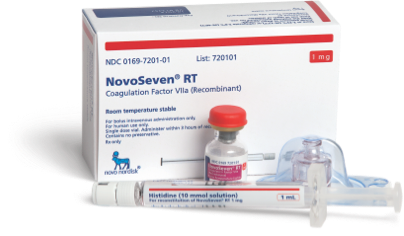
Help your patients make the switch to NovoSeven® RT. Learn more about Novo Nordisk’s trial prescription and support programs. To learn more about our trial prescription program, please call 1-844-668-6732 to speak with a NovoCare® Specialist.
bNovo Nordisk Hemophilia and Rare Bleeding Disorders Copay/Coinsurance Terms and Conditions:
Enrolled patients are eligible for up to $12,000 in co-pay/coinsurance assistance per calendar year for each NNI hemophilia or rare bleeding disorder product. Assistance is retroactive to 60 days. Patients must be commercially insured and may not be participating in any government, state, or federally funded medical or prescription benefit programs, including Medicare, Medicaid, Medigap, VA, DOD, and TRICARE, including patients who participate in a managed Medicaid program or have Medicaid as secondary insurance. Uninsured, cash-paying patients are not eligible to participate. Patients are eligible to receive co-pay/coinsurance assistance on an annual basis (12 months). Offer good only in the USA, Puerto Rico, Guam, Saipan, and Virgin Islands with participating pharmacies and cannot be redeemed at government-subsidized clinics. Void where taxed, restricted, or prohibited by law. The Savings Card is no longer valid for residents of Massachusetts. Patient is responsible for complying with any insurance carrier co-payment disclosure requirements, including disclosing any savings received from this program. Re-confirmation of information may be requested periodically to ensure accuracy of data and compliance with terms. This is not an insurance program. Novo Nordisk reserves the right to rescind, revoke, or amend this offer without notice at any time. Non-medication expenses, such as ancillary supplies or administration-related costs, are not eligible. Must have a current prescription for an FDA-approved indication.
cThe Novo Nordisk Patient Assistance Program (PAP) is administered by NovoCare® To qualify for the PAP, patients must demonstrate financial need and must have attempted to find alternative reimbursement. Several factors are considered in evaluating financial need, including cost of living, size of household, and burden of total medical expenses. If the applicant qualifies under the PAP guidelines, a limited supply of the requested medication(s) will be shipped to the patient. Patients who qualify for PAP may be eligible to receive the prescribed Novo Nordisk product, for up to 1 year from the approval date. Product limits vary.
dPatients who have been prescribed a Novo Nordisk hemophilia and rare bleeding disorder product for an FDA-approved indication, and who have commercial insurance, may be eligible to receive a limited supply of free product. Patients who participate in any government, state, or federally funded medical or prescription benefit programs, including Medicare, Medicaid, Medigap, VA, DOD, and TRICARE, including patients who participate in a managed Medicaid program or have Medicaid as secondary insurance, are not eligible to receive product support. Product is provided at no cost to the patient or the HCP, is not contingent on any product purchase, and the patient and HCP must not: (1) bill any third party for the free product, or (2) resell the free product.
Looking for other NovoSeven® RT resources?
Browse videos, podcasts, and more for professionals.

Help patients learn about bleeding disorders.
Important Safety Information for NovoSeven® RT
WARNING: THROMBOSIS
- Serious arterial and venous thrombotic events following administration of NovoSeven® RT have been reported
- Discuss the risks and explain the signs and symptoms of thrombotic and thromboembolic events to patients who will receive NovoSeven® RT
- Monitor patients for signs or symptoms of activation of the coagulation system and for thrombosis
Warnings and Precautions
- Serious arterial and venous thrombotic events have been reported in clinical trials and postmarketing surveillance
- Patients with congenital hemophilia receiving concomitant treatment with aPCCs (activated prothrombin complex concentrates), older patients particularly with acquired hemophilia and receiving other hemostatic agents, and patients with a history of cardiac and vascular disease may have an increased risk of developing thrombotic events
- Hypersensitivity reactions, including anaphylaxis, can occur with NovoSeven® RT. Patients with a known hypersensitivity to mouse, hamster, or bovine proteins may be at a higher risk of hypersensitivity reactions. Discontinue infusion and administer appropriate treatment when hypersensitivity reactions occur
- Factor VII deficient patients should be monitored for prothrombin time (PT) and factor VII coagulant activity (FVII:C). If FVII:C fails to reach the expected level, or PT is not corrected, or bleeding is not controlled after treatment with the recommended doses, antibody formation may be suspected and analysis for antibodies should be performed
- Laboratory coagulation parameters (PT/INR, aPTT, FVII:C) have shown no direct correlation to achieving hemostasis
Adverse Reactions
- The most common and serious adverse reactions in clinical trials are thrombotic events. Thrombotic adverse reactions following the administration of NovoSeven® RT in clinical trials occurred in 4% of patients with acquired hemophilia and 0.2% of bleeding episodes in patients with congenital hemophilia
Drug Interactions
- Thrombosis may occur if NovoSeven® RT is administered concomitantly with Coagulation Factor XIII
Please click here for NovoSeven® RT Prescribing Information, including Boxed Warning.
Indications and Usage
NovoSeven® RT (coagulation Factor VIIa, recombinant) is a coagulation factor indicated for:
- Treatment of bleeding episodes and perioperative management in adults and children with hemophilia A or B with inhibitors, congenital Factor VII (FVII) deficiency, and Glanzmann’s thrombasthenia with refractoriness to platelet transfusions, with or without antibodies to platelets
- Treatment of bleeding episodes and perioperative management in adults with acquired hemophilia
Important Safety Information for NovoSeven® RT
WARNING: THROMBOSIS
- Serious arterial and venous thrombotic events following administration of NovoSeven® RT have been reported
- Discuss the risks and explain the signs and symptoms of thrombotic and thromboembolic events to patients who will receive NovoSeven® RT
- Monitor patients for signs or symptoms of activation of the coagulation system and for thrombosis
Important Safety Information for NovoSeven® RT
WARNING: THROMBOSIS
- Serious arterial and venous thrombotic events following administration of NovoSeven® RT have been reported
- Discuss the risks and explain the signs and symptoms of thrombotic and thromboembolic events to patients who will receive NovoSeven® RT
- Monitor patients for signs or symptoms of activation of the coagulation system and for thrombosis
Warnings and Precautions
- Serious arterial and venous thrombotic events have been reported in clinical trials and postmarketing surveillance
- Patients with congenital hemophilia receiving concomitant treatment with aPCCs (activated prothrombin complex concentrates), older patients particularly with acquired hemophilia and receiving other hemostatic agents, and patients with a history of cardiac and vascular disease may have an increased risk of developing thrombotic events
- Hypersensitivity reactions, including anaphylaxis, can occur with NovoSeven® RT. Patients with a known hypersensitivity to mouse, hamster, or bovine proteins may be at a higher risk of hypersensitivity reactions. Discontinue infusion and administer appropriate treatment when hypersensitivity reactions occur
- Factor VII deficient patients should be monitored for prothrombin time (PT) and factor VII coagulant activity (FVII:C). If FVII:C fails to reach the expected level, or PT is not corrected, or bleeding is not controlled after treatment with the recommended doses, antibody formation may be suspected and analysis for antibodies should be performed
- Laboratory coagulation parameters (PT/INR, aPTT, FVII:C) have shown no direct correlation to achieving hemostasis
Adverse Reactions
- The most common and serious adverse reactions in clinical trials are thrombotic events. Thrombotic adverse reactions following the administration of NovoSeven® RT in clinical trials occurred in 4% of patients with acquired hemophilia and 0.2% of bleeding episodes in patients with congenital hemophilia
Drug Interactions
- Thrombosis may occur if NovoSeven® RT is administered concomitantly with Coagulation Factor XIII
Please click here for NovoSeven® RT Prescribing Information, including Boxed Warning.
Indications and Usage
NovoSeven® RT (coagulation Factor VIIa, recombinant) is a coagulation factor indicated for:
- Treatment of bleeding episodes and perioperative management in adults and children with hemophilia A or B with inhibitors, congenital Factor VII (FVII) deficiency, and Glanzmann’s thrombasthenia with refractoriness to platelet transfusions, with or without antibodies to platelets
- Treatment of bleeding episodes and perioperative management in adults with acquired hemophilia
