For chronic weight management as an adjunct to a reduced-calorie diet and increased physical activity for adults and pediatric patients aged 12 years and older with body weight greater than 60 kg and obesity, and for adults with overweight in the presence of at least one weight-related comorbid condition. Click for Limitations of Use.
Significant Weight Loss
In clinical trials, a majority of adult patients achieved clinically meaningful weight loss of ≥5% with Saxenda®1
Patients Lost Weight With Saxenda®
In a 1-year study in adults, 85% of patients treated with Saxenda® lost some weight1
In a 56-week study of 3,731 patients without type 2 diabetes and with a BMI ≥30 kg/m2, or ≥27% with at least 1 weight-related comorbidity, patients were randomized to either Saxenda® (n=2,487) or placebo (n=1,244), with all patients receiving a reduced-calorie diet (~500 kcal/day deficit) and physical activity counseling.1
The majority of patients achieved clinically meaningful weight loss of ≥5% with Saxenda®1
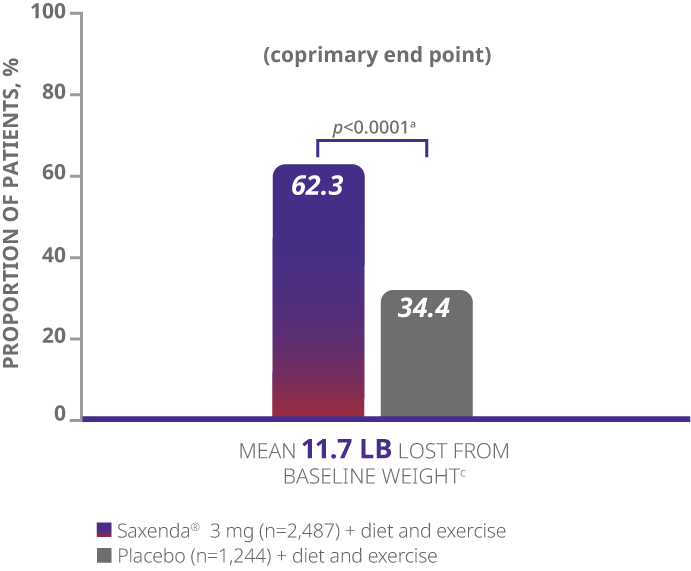
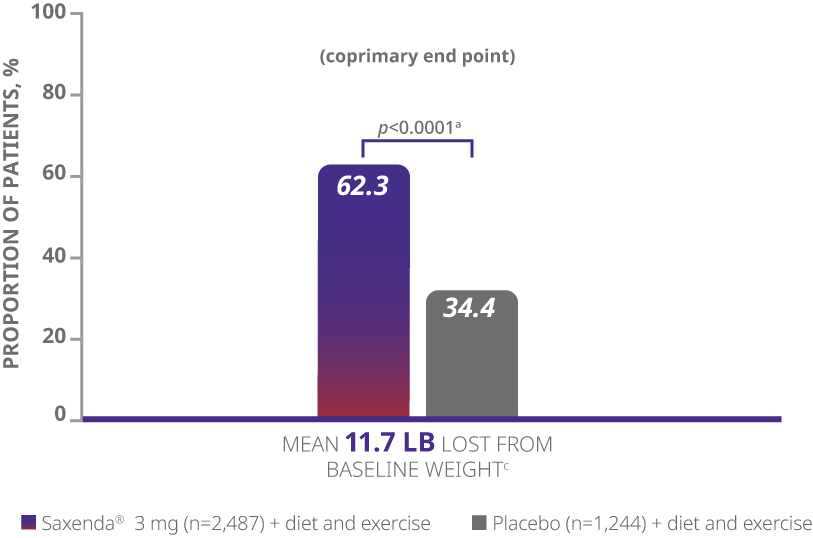
aDifference from placebo (least squares [LS] mean), 27.9% (95% CI, 23.9, 31.9).
bDifference from placebo (LS mean), 18.5% (95% CI, 15.2, 21.7).
cWeight loss in pounds (lb) calculated as 5%, 10%, or 20% of mean baseline body weight.
dBased on the frequency cumulative distribution of change in body weight curve.
- The primary end points were mean percentage weight change, percentage of patients achieving ≥5% of baseline weight loss, and percentage of patients achieving >10% of baseline weight loss1
- Weight loss was defined as any reduction in weight from start of trial1
- Mean baseline BMI was 38.3 kg/m2 and mean baseline body weight was 233.9 lb1
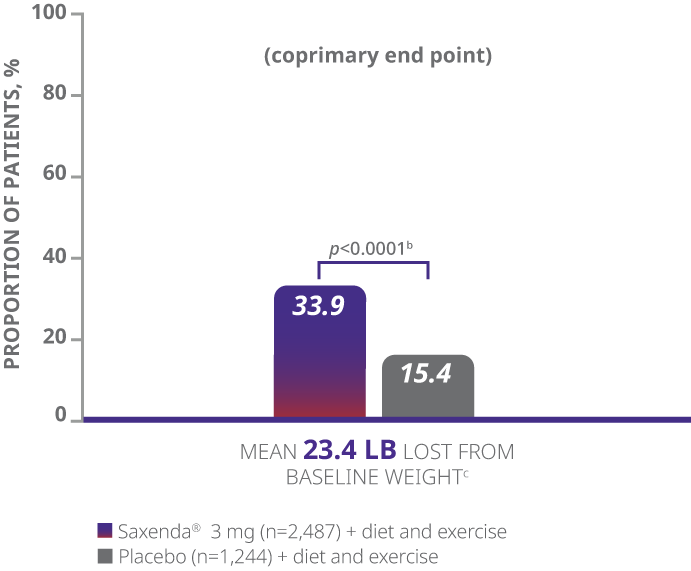
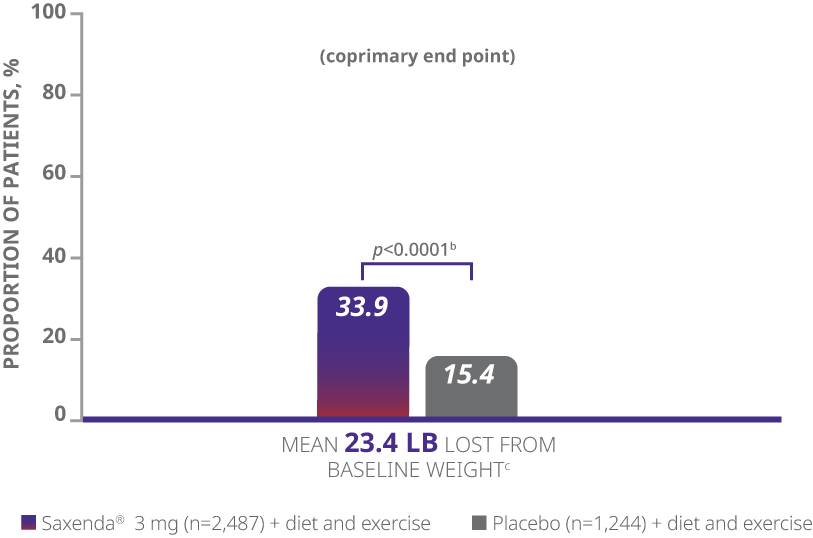
aDifference from placebo (least squares [LS] mean), 27.9% (95% CI, 23.9, 31.9).
bDifference from placebo (LS mean), 18.5% (95% CI, 15.2, 21.7).
cWeight loss in pounds (lb) calculated as 5%, 10%, or 20% of mean baseline body weight.
dBased on the frequency cumulative distribution of change in body weight curve.
- The primary end points were mean percentage weight change, percentage of patients achieving ≥5% of baseline weight loss, and percentage of patients achieving >10% of baseline weight loss1
- Weight loss was defined as any reduction in weight from start of trial1
- Mean baseline BMI was 38.3 kg/m2 and mean baseline body weight was 233.9 lb1
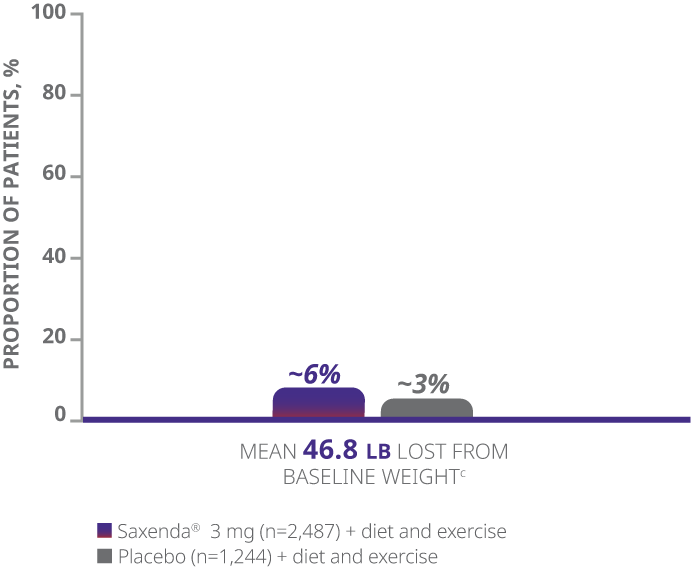

aDifference from placebo (least squares [LS] mean), 27.9% (95% CI, 23.9, 31.9).
bDifference from placebo (LS mean), 18.5% (95% CI, 15.2, 21.7).
cWeight loss in pounds (lb) calculated as 5%, 10%, or 20% of mean baseline body weight.
dBased on the frequency cumulative distribution of change in body weight curve.
- The primary end points were mean percentage weight change, percentage of patients achieving ≥5% of baseline weight loss, and percentage of patients achieving >10% of baseline weight loss1
- Weight loss was defined as any reduction in weight from start of trial1
- Mean baseline BMI was 38.3 kg/m2 and mean baseline body weight was 233.9 lb1
Clinically Meaningful Weight Loss
The majority of patients treated with Saxenda® achieved and maintained clinically meaningful weight loss1
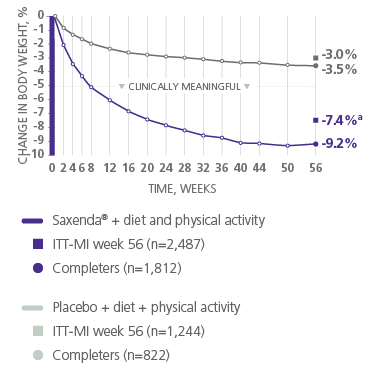
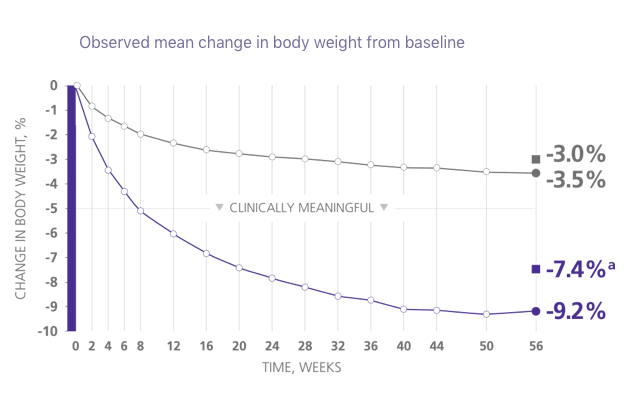

aDifference from placebo was statistically significant. ITT-MI, intention to treat with multiple imputations.1
21 lb mean weight loss
achieved by patients on Saxenda® who completed the study2
- Mean baseline body weight was 233.9 lb and mean baseline BMI was 38.3 kg/m2
Secondary End Point: Impact on Waist Circumference
A large waist circumference is an important marker for obesity-related health risks3
Measuring waist3
Measuring circumference correctly is simple to learn and important to know
Saxenda® reduced waist circumference by 3.2 in vs 1.6 in with placebo.1
Mean waist circumference at baseline was 45.3 inches for patients treated with Saxenda® (n=2,487) and 45.1 inches for patients taking placebo (n=1,244)1
Study 1 (1 year)1,4
- Results from a 56-week, randomized, double-blind, placebo-controlled study to evaluate the safety and efficacy of Saxenda®
- Adult patients with a BMI of ≥30, or ≥27 with 1 or more weight-related comorbidities (N=3,731) were randomized to receive once-daily Saxenda® (n=2,487) or placebo (n=1,244) in conjunction with a lifestyle modification program that included increased physical activity and a 500-kcal/day deficit diet
- Patients underwent a 4-week dose-escalation period followed by 52 weeks on the full dose
- The primary end points were mean percent weight change, percentage of patients achieving ≥5% of baseline weight loss, and percentage of patients achieving >10% of baseline weight loss at 56 weeks
- Secondary end points included changes in waist circumference, blood pressure, and lipids
- Mean baseline body weight was 233.9 lb and mean BMI was 38.3 kg/m2
- Patients with type 2 diabetes were excluded from participating
RECOMMENDED CONTENT
Sustained Weight Loss
Important Safety Information for Saxenda®
WARNING: RISK OF THYROID C-CELL TUMORS
Liraglutide causes dose-dependent and treatment-duration-dependent thyroid C-cell tumors at clinically relevant exposures in both genders of rats and mice. It is unknown whether Saxenda® causes thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans, as the human relevance of liraglutide-induced rodent thyroid C-cell tumors has not been determined.
Saxenda® is contraindicated in patients with a personal or family history of MTC and in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Counsel patients regarding the potential risk of MTC with use of Saxenda® and inform them of symptoms of thyroid tumors (eg, a mass in the neck, dysphagia, dyspnea, persistent hoarseness). Routine monitoring of serum calcitonin or using thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with Saxenda®.
Contraindications
Saxenda® is contraindicated in:
- Patients with a personal or family history of MTC or patients with MEN 2
- Patients with a serious hypersensitivity reaction to liraglutide or to any of the excipients in Saxenda®. Serious hypersensitivity reactions including anaphylactic reactions and angioedema have been reported with Saxenda®
Warnings and Precautions
- Risk of Thyroid C-cell Tumors: If serum calcitonin is measured and found to be elevated, the patient should be further evaluated. Patients with thyroid nodules noted on physical examination or neck imaging should also be further evaluated
- Acute Pancreatitis: Acute pancreatitis, including fatal and non-fatal hemorrhagic or necrotizing pancreatitis, has been observed in patients treated with GLP-1 receptor agonists, including liraglutide. Observe patients carefully for signs and symptoms of acute pancreatitis, which may include persistent or severe abdominal pain (sometimes radiating to the back), nausea or vomiting. If pancreatitis is suspected, discontinue Saxenda® and initiate appropriate management
- Acute Gallbladder Disease: Substantial or rapid weight loss can increase the risk of cholelithiasis; however, the incidence of acute gallbladder disease was greater in patients treated with Saxenda® than with placebo even after accounting for the degree of weight loss. If cholelithiasis is suspected, gallbladder studies and appropriate clinical follow-up are indicated
- Hypoglycemia: Adult patients with type 2 diabetes on an insulin secretagogue (eg, a sulfonylurea) or insulin may have an increased risk of hypoglycemia, including severe hypoglycemia with use of Saxenda®. The risk may be lowered by a reduction in the dose of insulin secretagogues or insulin. In pediatric patients without type 2 diabetes, hypoglycemia occurred. Inform all patients of the risk of hypoglycemia and educate them on the signs and symptoms
- Heart Rate Increase: Mean increases in resting heart rate of 2 to 3 beats per minute (bpm) were observed in patients treated with Saxenda®. Monitor heart rate at regular intervals and inform patients to report palpitations or feelings of a racing heartbeat while at rest during treatment with Saxenda®. Discontinue Saxenda® in patients who experience a sustained increase in resting heart rate
- Acute Kidney Injury Due to Volume Depletion: There have been postmarketing reports of acute kidney injury which may sometimes require hemodialysis, in patients treated with liraglutide. The majority of the reported events occurred in patients who had experienced gastrointestinal reactions leading to dehydration such as nausea, vomiting, or diarrhea. Monitor renal function in patients reporting adverse reactions to Saxenda® that could lead to volume depletion, especially during initation and escalation of Saxenda®
- Severe Gastrointestinal Adverse Reactions: Use of Saxenda® has been associated with gastrointestinal adverse reactions, sometimes severe. In clinical trials, severe gastrointestinal adverse reactions were reported more frequently among patients receiving Saxenda® (4.8%) than placebo (1.4%). Saxenda® is not recommended in patients with severe gastroparesis
- Hypersensitivity Reactions: Serious hypersensitivity reactions (eg, anaphylaxis and angioedema) have been reported in patients treated with Saxenda®. If a hypersensitivity reaction occurs, patients should stop taking Saxenda® and promptly seek medical advice
- Pulmonary Aspiration During General Anesthesia or Deep Sedation: Saxenda® delays gastric emptying. There have been rare postmarketing reports of pulmonary aspiration in patients receiving GLP-1 receptor agonists undergoing elective surgeries or procedures requiring general anesthesia or deep sedation who had residual gastric contents despite reported adherence to preoperative fasting recommendations. Instruct patients to inform healthcare providers prior to any planned surgeries or procedures if they are taking Saxenda®
Adverse Reactions
- The most common adverse reactions, reported in ≥5% are nausea, diarrhea, constipation, vomiting, injection site reactions, headache, hypoglycemia, dyspepsia, fatigue, dizziness, abdominal pain, increased lipase, upper abdominal pain, pyrexia, and gastroenteritis
Drug Interactions
- Saxenda® causes a delay of gastric emptying and has the potential to impact the absorption of concomitantly administered oral medications. Monitor for potential consequences of delayed absorption of oral medications concomitantly administered with Saxenda®
Use in Specific Populations
- Saxenda® should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus
- There are no data on the presence of liraglutide in human breast milk; liraglutide was present in the milk of lactating rats
- Saxenda® has not been studied in patients less than 12 years of age
Please click here for Saxenda® Prescribing Information, including Boxed Warning.
Indications and Usage
Saxenda® (liraglutide) injection 3 mg is indicated in combination with reduced-calorie diet and increased physical activity to reduce excess body weight and maintain weight reduction long term in:
- Adults and pediatric patients aged 12 years and older with body weight greater than 60kg and obesity
- Adults with overweight in the presence of at least one weight related comorbid condition
Limitations of Use
- Saxenda® contains liraglutide and should not be coadministered with other liraglutide-containing products or with any other GLP-1 receptor agonist
- The safety and effectiveness of Saxenda® in pediatric patients with type 2 diabetes have not been established
Important Safety Information for Saxenda®
WARNING: RISK OF THYROID C-CELL TUMORS
Liraglutide causes dose-dependent and treatment-duration-dependent thyroid C-cell tumors at clinically relevant exposures in both genders of rats and mice. It is unknown whether Saxenda® causes thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans, as the human relevance of liraglutide-induced rodent thyroid C-cell tumors has not been determined.
Saxenda® is contraindicated in patients with a personal or family history of MTC and in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Counsel patients regarding the potential risk of MTC with use of Saxenda® and inform them of symptoms of thyroid tumors (eg, a mass in the neck, dysphagia, dyspnea, persistent hoarseness). Routine monitoring of serum calcitonin or using thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with Saxenda®.
Important Safety Information for Saxenda®
WARNING: RISK OF THYROID C-CELL TUMORS
Liraglutide causes dose-dependent and treatment-duration-dependent thyroid C-cell tumors at clinically relevant exposures in both genders of rats and mice. It is unknown whether Saxenda® causes thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans, as the human relevance of liraglutide-induced rodent thyroid C-cell tumors has not been determined.
Saxenda® is contraindicated in patients with a personal or family history of MTC and in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Counsel patients regarding the potential risk of MTC with use of Saxenda® and inform them of symptoms of thyroid tumors (eg, a mass in the neck, dysphagia, dyspnea, persistent hoarseness). Routine monitoring of serum calcitonin or using thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with Saxenda®.
Contraindications
Saxenda® is contraindicated in:
- Patients with a personal or family history of MTC or patients with MEN 2
- Patients with a serious hypersensitivity reaction to liraglutide or to any of the excipients in Saxenda®. Serious hypersensitivity reactions including anaphylactic reactions and angioedema have been reported with Saxenda®
Warnings and Precautions
- Risk of Thyroid C-cell Tumors: If serum calcitonin is measured and found to be elevated, the patient should be further evaluated. Patients with thyroid nodules noted on physical examination or neck imaging should also be further evaluated
- Acute Pancreatitis: Acute pancreatitis, including fatal and non-fatal hemorrhagic or necrotizing pancreatitis, has been observed in patients treated with GLP-1 receptor agonists, including liraglutide. Observe patients carefully for signs and symptoms of acute pancreatitis, which may include persistent or severe abdominal pain (sometimes radiating to the back), nausea or vomiting. If pancreatitis is suspected, discontinue Saxenda® and initiate appropriate management
- Acute Gallbladder Disease: Substantial or rapid weight loss can increase the risk of cholelithiasis; however, the incidence of acute gallbladder disease was greater in patients treated with Saxenda® than with placebo even after accounting for the degree of weight loss. If cholelithiasis is suspected, gallbladder studies and appropriate clinical follow-up are indicated
- Hypoglycemia: Adult patients with type 2 diabetes on an insulin secretagogue (eg, a sulfonylurea) or insulin may have an increased risk of hypoglycemia, including severe hypoglycemia with use of Saxenda®. The risk may be lowered by a reduction in the dose of insulin secretagogues or insulin. In pediatric patients without type 2 diabetes, hypoglycemia occurred. Inform all patients of the risk of hypoglycemia and educate them on the signs and symptoms
- Heart Rate Increase: Mean increases in resting heart rate of 2 to 3 beats per minute (bpm) were observed in patients treated with Saxenda®. Monitor heart rate at regular intervals and inform patients to report palpitations or feelings of a racing heartbeat while at rest during treatment with Saxenda®. Discontinue Saxenda® in patients who experience a sustained increase in resting heart rate
- Acute Kidney Injury Due to Volume Depletion: There have been postmarketing reports of acute kidney injury which may sometimes require hemodialysis, in patients treated with liraglutide. The majority of the reported events occurred in patients who had experienced gastrointestinal reactions leading to dehydration such as nausea, vomiting, or diarrhea. Monitor renal function in patients reporting adverse reactions to Saxenda® that could lead to volume depletion, especially during initation and escalation of Saxenda®
- Severe Gastrointestinal Adverse Reactions: Use of Saxenda® has been associated with gastrointestinal adverse reactions, sometimes severe. In clinical trials, severe gastrointestinal adverse reactions were reported more frequently among patients receiving Saxenda® (4.8%) than placebo (1.4%). Saxenda® is not recommended in patients with severe gastroparesis
- Hypersensitivity Reactions: Serious hypersensitivity reactions (eg, anaphylaxis and angioedema) have been reported in patients treated with Saxenda®. If a hypersensitivity reaction occurs, patients should stop taking Saxenda® and promptly seek medical advice
- Pulmonary Aspiration During General Anesthesia or Deep Sedation: Saxenda® delays gastric emptying. There have been rare postmarketing reports of pulmonary aspiration in patients receiving GLP-1 receptor agonists undergoing elective surgeries or procedures requiring general anesthesia or deep sedation who had residual gastric contents despite reported adherence to preoperative fasting recommendations. Instruct patients to inform healthcare providers prior to any planned surgeries or procedures if they are taking Saxenda®
Adverse Reactions
- The most common adverse reactions, reported in ≥5% are nausea, diarrhea, constipation, vomiting, injection site reactions, headache, hypoglycemia, dyspepsia, fatigue, dizziness, abdominal pain, increased lipase, upper abdominal pain, pyrexia, and gastroenteritis
Drug Interactions
- Saxenda® causes a delay of gastric emptying and has the potential to impact the absorption of concomitantly administered oral medications. Monitor for potential consequences of delayed absorption of oral medications concomitantly administered with Saxenda®
Use in Specific Populations
- Saxenda® should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus
- There are no data on the presence of liraglutide in human breast milk; liraglutide was present in the milk of lactating rats
- Saxenda® has not been studied in patients less than 12 years of age
Please click here for Saxenda® Prescribing Information, including Boxed Warning.
Indications and Usage
Saxenda® (liraglutide) injection 3 mg is indicated in combination with reduced-calorie diet and increased physical activity to reduce excess body weight and maintain weight reduction long term in:
- Adults and pediatric patients aged 12 years and older with body weight greater than 60kg and obesity
- Adults with overweight in the presence of at least one weight related comorbid condition
Limitations of Use
- Saxenda® contains liraglutide and should not be coadministered with other liraglutide-containing products or with any other GLP-1 receptor agonist
- The safety and effectiveness of Saxenda® in pediatric patients with type 2 diabetes have not been established
References
1. Saxenda® [package insert]. Plainsboro, NJ: Novo Nordisk Inc.
2. Data on file. Novo Nordisk Inc.; Plainsboro, NJ.
3. National Heart, Lung, and Blood Institute; National Institutes of Health; North American Association for the Study of Obesity. The Practical Guide: Identification, Evaluation, and Treatment of Overweight and Obesity in Adults. Bethesda, MD: National Institutes of Health; 2000. NIH Publication 00-4084.
4. Pi-Sunyer X, Astrup A, Fujioka K, et al. A randomized, controlled trial of 3.0 mg of liraglutide in weight management. N Engl J Med. 2015;373(1):11-22.