Sogroya® is indicated for the replacement of endogenous growth hormone in adults with growth hormone deficiency (GHD) and for the treatment of pediatric patients aged 2.5 years and older with: growth failure due to inadequate secretion of endogenous growth hormone (GH), short stature born Small for Gestational Age (SGA) and with no catch-up growth by 2 years of age, growth failure associated with Noonan Syndrome (NS), and Idiopathic Short Stature (ISS). Please see full indications.
EFFICACY AND SAFETY DATA
for pediatric patients 2.5 years and older with other specific growth-related conditions
with short stature born Small for Gestational Age (SGA) and with no catch-up growth by 2 years of age, growth failure associated with Noonan Syndrome (NS), and Idiopathic Short Stature (ISS).
Actor portrayal
Pediatric patients (2.5 and older) with
Idiopathic Short Stature (ISS)
Sogroya® demonstrated similar efficacy to daily GH treatment, with fewer injections1
REAL8 main phase: Primary endpoint AHV at Week 521,2
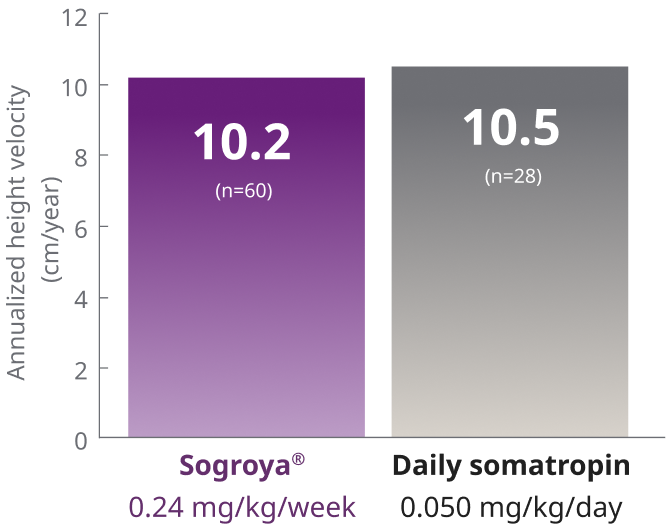
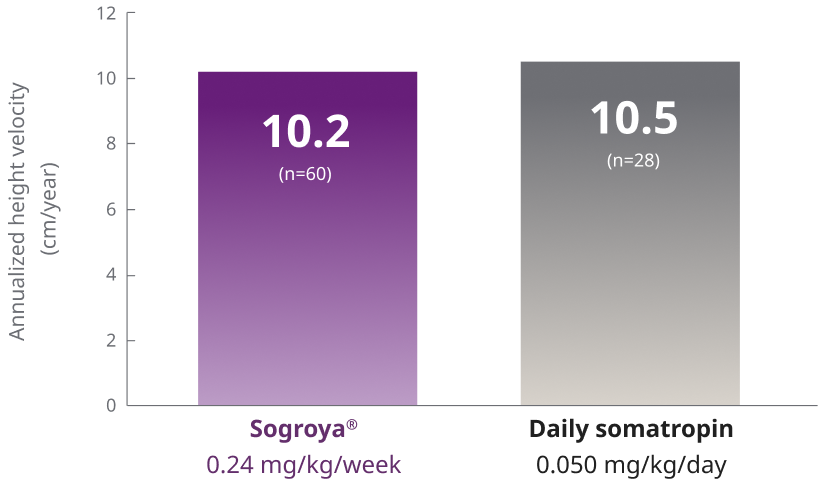
ETD (95% CI) = –0.3 (–1.0; 0.42)
Recommended dosage of Sogroya® for ISS2


REAL8 was a global, multicenter, randomized, open-label, active-comparator, phase 3 basket study. A total of 88 GH-treatment-naïve children 2.8-10.8 years with ISS were randomized to Sogroya® 0.24 mg/kg/week (n=60) or daily somatropin 0.05 mg/kg/day (n=28). Dose 0.05 mg/kg/day of daily somatropin is less than the maximum dose (0.067 mg/kg/day) approved for use in pediatric patients with ISS in the US. The primary endpoint was AHV at Week 52.1
AHV=annualized height velocity; CI=confidence interval; ETD=estimated treatment difference; GH=growth hormone; ISS=Idiopathic Short Stature.
Proven safety profile1
REAL8 main phase: Adverse reactions occurring ≥10% in Sogroya® or somatropin-treated pediatric patients (52 weeks)1
aRespiratory tract infection in the Sogroya® treatment group included influenza (16.9%), upper respiratory tract infection (6.8%), respiratory tract infection (3.4%), pneumonia (1.7%), pneumonia bacterial (1.7%), respiratory tract infection viral (1.7%), and viral upper respiratory tract infection (1.7%).
bNasopharyngitis in the Sogroya® treatment group included nasopharyngitis (15.3%), pharyngitis streptococcal (5.1%), pharyngitis (1.7%), rhinitis (1.7%), sinusitis (1.7%), and tracheitis (1.7%).
cEar infection in the Sogroya® treatment group included otitis media (6.8%), ear infection (3.4%), and otitis media acute (1.7%).
dDiarrhea in the Sogroya® treatment group included gastroenteritis (3.4%), gastroenteritis viral (3.4%), diarrhea (1.7%), enterobiasis (1.7%), and parasitic gastroenteritis (1.7%).
eInjection-site reaction in the Sogroya® treatment group included application-site reaction (1.7%), injection-site bruising (1.7%), injection-site hematoma (1.7%), injection-site hemorrhage (1.7%), injection-site pruritis (1.7%), a n d injection-site urticaria (1.7%).
fCough in the Sogroya® treatment group included cough (5.1%).
gVomiting in the Sogroya® treatment group included vomiting (5.1%).
ISI=Idiopathic Short Stature.
REAL8: A global, multicenter, randomized, open-labeled, active-comparator, phase 3 basket study1
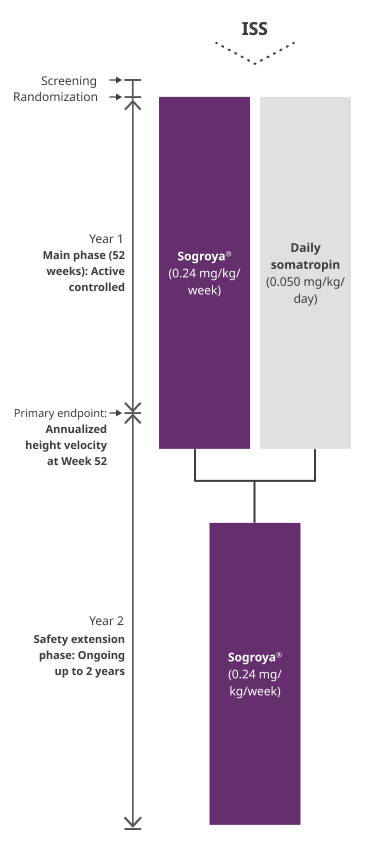
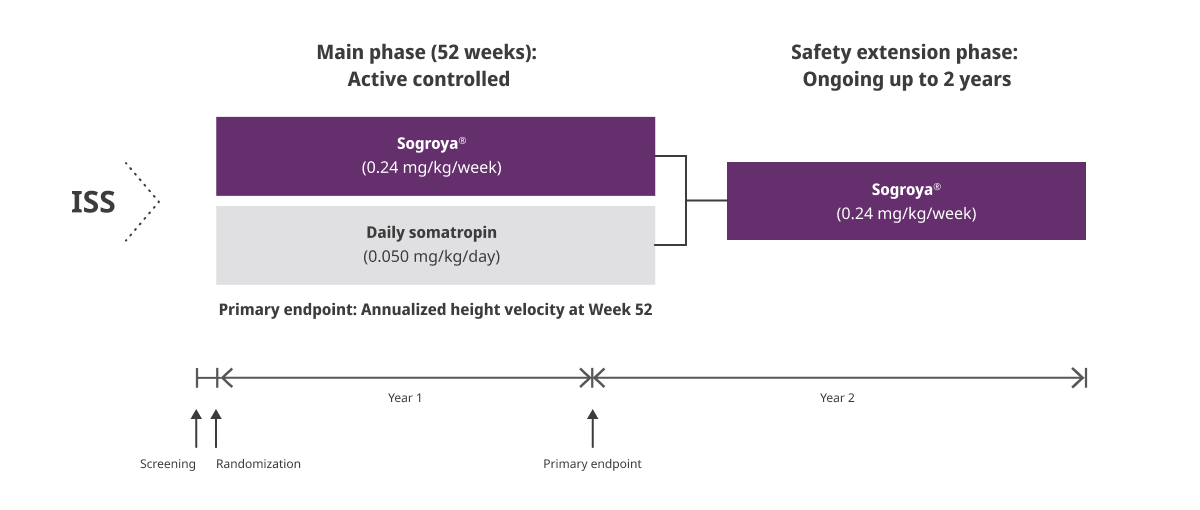
ISS= Idiopathic Short Stature.
Pediatric patients (2.5 and older) born
Small for Gestational Age (SGA)
with no catch-up growth by 2 years of age
Sogroya®: Non-inferior efficacy with fewer injections compared with daily GH1
REAL8 main phase: Primary endpoint AHV at Week 521,2
Recommended dosage of Sogroya® for SGA2


REAL8 was a global, multicenter, randomized, open-label, active-comparator, phase3 basket study. A total of 142 GH-treatment-naïve children 2.6-10.7 years born SGA were randomized to Sogroya® 0.24 mg/kg/week (n=70) daily somatropin 0.035 mg/kg/day (n=37), or daily somatropin 0.067 mg/kg/day (n=35). Dose 0.035 mg/kg/day of daily somatropin is less than the maximum dose (0.067 mg/kg/day) approved for use in pediatric patients with SGA in the US. The primary endpoint was AHV at Week 52.1
AHV=annualized height velocity; CI=confidence interval; ETD=estimated treatment difference; GH=growth hormone; IGF-1=insulin-like growth factor; SGA=Small for Gestational Age.
Proven safety profile1
REAL8 main phase: Adverse reactions occurring ≥10% in Sogroya® or somatropin-treated pediatric patients (52 weeks)1
hNasopharyngitis in the Sogroya® treatment group included nasopharyngitis (15.9%), pharyngitis (2.9%), rhinitis (2.9%), pharyngitis streptococcal (1.4%), sinusitis (1.4%), and viral rhinitis (1.4%).
iRespiratory tract infections in the Sogroya® treatment group included upper respiratory tract infection (20.3%), influenza (10.1 %), metapneuomovirus infection (1.4%), mycoplasma infection (1.4%), pneumonia (1.4%), pneumonia bacterial (1.4%), pneumonia mycoplasmal (1.4%), respiratory syncytial virus infection (1.4%), and respiratory tract infection (1.4%).
jCough in the Sogroya® treatment group included cough (15.9%).
kEar infection in the Sogroya® treatment group included otitis media (7.2%), ear infection (2.9%), otitis externa (1.4%), and otitis media acute (1.4%).
lDiarrhea in the Sogroya® treatment group included diarrhea (4.3%), gastroenteritis (4.3%), and gastrointestinal viral infection (1.4%).
mVomiting in the Sogroya® treatment group included vomiting (10.1%).
SGA=Small for Gestational Age.
REAL8: A global, multicenter, randomized, open-labeled, active-comparator, phase 3 basket study1
SGA=small for gestational age.
Pediatric patients (2.5 and older) with
Noonan Syndrome (NS)
Sogroya® demonstrated similar growth with fewer injections when compared with daily GH1
REAL8 main phase: Primary endpoint AHV at Week 521,2
ETD (95% CI) = 1.2 (0.32; 2.03)
Recommended dosage of Sogroya® for NS2


REAL8 was a global, multicenter, randomized, open-label, active-comparator, phase 3 basket study. A total of 77 GH-treatment-naïve children 2-11.1 years with NS were randomized to Sogroya® 0.24 mg/kg/week (n=49) or daily somatropin 0.05 mg/kg/day (n=28). Dose 0.05 mg/kg/day of daily somatropin is less than the maximum dose (0.066 mg/kg/day) approved for use in pediatric patients with NS in the United States. The primary endpoint was AHV at Week 52.1
AHV=annualized height velocity; CI=confidence interval; ETD=estimated treatment difference; GH=growth hormone; NS=Noonan Syndrome.
Proven safety profile1
REAL8 main phase: Adverse reactions occurring ≥10% in Sogroya® or somatropin-treated pediatric patients (52 weeks)1
nRespiratory tract infection in the Sogroya® treatment group included upper respiratory tract infection (20.4%), influenza (10.2%), pneumonia (6.1 %), influenza like illness (2%), respiratory syncytial virus infection (2%), respiratory tract infection (2%), respiratory tract infection viral (2%), upper respiratory tract infection bacterial (2%), and viral upper respiratory tract infection (2%).
oNasopharyngitis in the Sogroya® treatment group included nasopharyngitis (24.5%), pharyngitis (4.1 %), bacterial infection (2%), herpes pharyngitis (2%), pharyngitis streptococcal (2%), pharyngotonsillitis (2%), and rhinitis (2%).
pDiarrhea in the Sogroya® treatment group included gastroenteritis (14.3%), diarrhea (6.1%), gastroenteritis viral (2%), and parasitic gastroenteritis (2%).
qEar infection in the Sogroya® treatment group included otitis media (8.2%), otitis externa (4.1 %), ear infection (2%), otitis media acute (2%), and otitis media chronic (2%).
rCough in the Sogroya® treatment group included cough (12.2%) and bacterial infection (2%).
sVomiting in the Sogroya® treatment group included vomiting (8.2%) and gastritis (4.1%).
tInjection site reaction in the Sogroya® treatment group included injection site bruising (6.1%) and injection site hemorrhage (2%).
uAbdominal pain in the SOGROYA treatment group included abdominal distension (2%).
NS=Noonan Syndrome.
REAL8: A global, multicenter, randomized, open-labeled, active-comparator, phase 3 basket study1
NS=Noonan Syndrome.
Other specific growth-related conditions: IGF-1 Response
Explore IGF-1 response for pediatric patients with other specific growth-related conditions.
Dosing for pediatric patients
See dosing information for switch and treatment-naïve patients.
Important Safety Information for Sogroya®
Contraindications
Sogroya® is contraindicated in patients with:
- acute critical illness after open-heart surgery, abdominal surgery, multiple accidental trauma, or acute respiratory failure because of the risk of increased mortality with use of Sogroya®
- hypersensitivity to Sogroya® or any of its excipients. Systemic hypersensitivity reactions have been reported postmarketing with somatropin
- pediatric patients with closed epiphyses
- active malignancy
- active proliferative or severe non-proliferative diabetic retinopathy
- pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to risk of sudden death
Warnings & Precautions
- Increased Mortality in Patients with Acute Critical Illness: Increased mortality has been reported after treatment with somatropin in patients with acute critical illness due to complications following open-heart surgery, abdominal surgery, multiple accidental trauma, and in patients with acute respiratory failure
- Severe Hypersensitivity: Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported postmarketing with use of somatropin. Inform patients and/or caregivers that such reactions are possible and that prompt medical attention should be sought if an allergic reaction occurs
- Increased Risk of Neoplasms: There is an increased risk of malignancy progression with somatropin in patients with active malignancy. Any preexisting malignancy should be inactive, and its treatment complete prior to instituting Sogroya®. In childhood cancer survivors treated with radiation to the brain/head for their first neoplasm who developed subsequent GHD and were treated with somatropin, an increased risk of a second neoplasm has been reported. Monitor patients with a history of GHD secondary to an intracranial neoplasm for progression or recurrence of the tumor. Children with certain rare genetic causes of short stature have an increased risk of developing malignancies and should be carefully monitored for development of neoplasms. Monitor patients for increased growth or potential malignant changes of preexisting nevi. Advise patients/caregivers to report changes in the appearance of preexisting nevi
- Glucose Intolerance and Diabetes Mellitus: Treatment with somatropin may decrease insulin sensitivity, particularly at higher doses. New onset type 2 diabetes has been reported. Monitor glucose levels in all patients, especially in those with existing diabetes mellitus or with risk factors for diabetes mellitus, such as obesity, Turner syndrome, or a family history of diabetes mellitus. The doses of antidiabetic agents may require adjustment when Sogroya® is initiated
- Intracranial Hypertension: Has been reported usually within 8 weeks of treatment initiation. Perform fundoscopic examination prior to initiation of treatment and periodically thereafter. If papilledema is identified, evaluate the etiology, and treat the underlying cause before initiating Sogroya®. If papilledema is observed, stop treatment. If intracranial hypertension is confirmed, Sogroya® can be restarted at a lower dose after intracranial hypertension signs and symptoms have resolved
- Fluid Retention: Was observed during Sogroya® therapy. Clinical manifestations of fluid retention (e.g. edema and nerve compression syndromes, including carpal tunnel syndrome/paresthesia) are usually transient and dose dependent
- Hypoadrenalism: Patients receiving somatropin therapy who have or are at risk for corticotropin deficiency may be at risk for reduced serum cortisol levels and/or unmasking of central (secondary) hypoadrenalism. Patients treated with glucocorticoid replacement for previously diagnosed hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®. Monitor patients with known hypoadrenalism for reduced serum cortisol levels and/or need for glucocorticoid dose increases
- Hypothyroidism: Undiagnosed/untreated hypothyroidism may prevent an optimal response to Sogroya®. Monitor thyroid function periodically as hypothyroidism may occur or worsen after initiation of Sogroya®
- Slipped Capital Femoral Epiphysis in Pediatric Patients: Slipped capital femoral epiphysis may occur more frequently in patients undergoing rapid growth. Slipped capital femoral epiphysis may lead to osteonecrosis. Cases of slipped capital femoral epiphysis with or without osteonecrosis have been reported in pediatric patients with short stature receiving somatropin. Evaluate pediatric patients receiving Sogroya® with the onset of a limp or complaints of persistent hip or knee pain for slipped capital femoral epiphysis and osteonecrosis and manage accordingly
- Progression of Preexisting Scoliosis in Pediatric Patients: Monitor patients with a history of scoliosis for disease progression
- Pancreatitis: Cases of pancreatitis have been reported in patients receiving somatropin. The risk may be greater in pediatric patients compared to adults. Consider pancreatitis in patients with persistent severe abdominal pain
- Lipohypertrophy/Lipoatrophy: May occur if Sogroya® is administered at the same site over a long period of time. Rotate injection sites to reduce this risk
- Sudden Death in Pediatric Patients with Prader-Willi Syndrome: There have been reports of fatalities after initiating therapy with somatropin in pediatric patients with Prader-Willi syndrome who had one or more of the following risk factors: severe obesity, history of upper airway obstruction or sleep apnea, or unidentified respiratory infection. Male patients with one or more of these factors may be at greater risk than females. Sogroya® is not indicated for the treatment of pediatric patients who have growth failure due to genetically confirmed Prader-Willi syndrome
- Laboratory Tests: Serum levels of inorganic phosphorus and alkaline phosphatase have increased after Sogroya® therapy. Serum levels of parathyroid hormone may increase with somatropin treatment
Adverse Reactions
- Common adverse reactions in pediatric patients treated with Sogroya® include: cough, diarrhea, ear infection, headache, injection site reaction, nasopharyngitis, pain in extremity, pyrexia, respiratory tract infection, and vomiting
- Adult patients with GHD: Adverse reactions reported in >2% of patients are: back pain, arthralgia, dyspepsia, sleep disorder, dizziness, tonsillitis, peripheral edema, vomiting, adrenal insufficiency, hypertension, blood creatine phosphokinase increase, weight increase, anemia
Drug Interactions
- Glucocorticoids: Patients treated with glucocorticoid for hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®
- Cytochrome P450-Metabolized Drugs: Sogroya® may alter the clearance. Monitor carefully if used with Sogroya®
- Oral Estrogen: Patients receiving oral estrogen replacement may require higher Sogroya® dosages
- Insulin and/or Other Antihyperglycemic Agents: Dose adjustment of insulin and/or antihyperglycemic agent may be required for patients with diabetes mellitus
Please click here for Sogroya® Prescribing Information.
Indications and Usage
Sogroya® (somapacitan-beco) injection 5 mg, 10 mg, or 15 mg is indicated for the:
- treatment of pediatric patients aged 2.5 years and older with:
- Growth failure due to inadequate secretion of endogenous growth hormone (GH)
- Short stature born Small for Gestational Age (SGA) and with no catch-up growth by 2 years of age
- Growth failure associated with Noonan Syndrome (NS)
- Idiopathic Short Stature (ISS)
- replacement of endogenous GH in adults with growth hormone deficiency (GHD)
Indications and Usage
Sogroya® (somapacitan-beco) injection 5 mg, 10 mg, or 15 mg is indicated for the:
- treatment of pediatric patients aged 2.5 years and older with:
- Growth failure due to inadequate secretion of endogenous growth hormone (GH)
- Short stature born Small for Gestational Age (SGA) and with no catch-up growth by 2 years of age
- Growth failure associated with Noonan Syndrome (NS)
- Idiopathic Short Stature (ISS)
- replacement of endogenous GH in adults with growth hormone deficiency (GHD)
Important Safety Information for Sogroya®
Contraindications
Sogroya® is contraindicated in patients with:
- acute critical illness after open-heart surgery, abdominal surgery, multiple accidental trauma, or acute respiratory failure because of the risk of increased mortality with use of Sogroya®
- hypersensitivity to Sogroya® or any of its excipients. Systemic hypersensitivity reactions have been reported postmarketing with somatropin
- pediatric patients with closed epiphyses
- active malignancy
- active proliferative or severe non-proliferative diabetic retinopathy
- pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to risk of sudden death
Warnings & Precautions
- Increased Mortality in Patients with Acute Critical Illness: Increased mortality has been reported after treatment with somatropin in patients with acute critical illness due to complications following open-heart surgery, abdominal surgery, multiple accidental trauma, and in patients with acute respiratory failure
- Severe Hypersensitivity: Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported postmarketing with use of somatropin. Inform patients and/or caregivers that such reactions are possible and that prompt medical attention should be sought if an allergic reaction occurs
- Increased Risk of Neoplasms: There is an increased risk of malignancy progression with somatropin in patients with active malignancy. Any preexisting malignancy should be inactive, and its treatment complete prior to instituting Sogroya®. In childhood cancer survivors treated with radiation to the brain/head for their first neoplasm who developed subsequent GHD and were treated with somatropin, an increased risk of a second neoplasm has been reported. Monitor patients with a history of GHD secondary to an intracranial neoplasm for progression or recurrence of the tumor. Children with certain rare genetic causes of short stature have an increased risk of developing malignancies and should be carefully monitored for development of neoplasms. Monitor patients for increased growth or potential malignant changes of preexisting nevi. Advise patients/caregivers to report changes in the appearance of preexisting nevi
- Glucose Intolerance and Diabetes Mellitus: Treatment with somatropin may decrease insulin sensitivity, particularly at higher doses. New onset type 2 diabetes has been reported. Monitor glucose levels in all patients, especially in those with existing diabetes mellitus or with risk factors for diabetes mellitus, such as obesity, Turner syndrome, or a family history of diabetes mellitus. The doses of antidiabetic agents may require adjustment when Sogroya® is initiated
- Intracranial Hypertension: Has been reported usually within 8 weeks of treatment initiation. Perform fundoscopic examination prior to initiation of treatment and periodically thereafter. If papilledema is identified, evaluate the etiology, and treat the underlying cause before initiating Sogroya®. If papilledema is observed, stop treatment. If intracranial hypertension is confirmed, Sogroya® can be restarted at a lower dose after intracranial hypertension signs and symptoms have resolved
- Fluid Retention: Was observed during Sogroya® therapy. Clinical manifestations of fluid retention (e.g. edema and nerve compression syndromes, including carpal tunnel syndrome/paresthesia) are usually transient and dose dependent
- Hypoadrenalism: Patients receiving somatropin therapy who have or are at risk for corticotropin deficiency may be at risk for reduced serum cortisol levels and/or unmasking of central (secondary) hypoadrenalism. Patients treated with glucocorticoid replacement for previously diagnosed hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®. Monitor patients with known hypoadrenalism for reduced serum cortisol levels and/or need for glucocorticoid dose increases
- Hypothyroidism: Undiagnosed/untreated hypothyroidism may prevent an optimal response to Sogroya®. Monitor thyroid function periodically as hypothyroidism may occur or worsen after initiation of Sogroya®
- Slipped Capital Femoral Epiphysis in Pediatric Patients: Slipped capital femoral epiphysis may occur more frequently in patients undergoing rapid growth. Slipped capital femoral epiphysis may lead to osteonecrosis. Cases of slipped capital femoral epiphysis with or without osteonecrosis have been reported in pediatric patients with short stature receiving somatropin. Evaluate pediatric patients receiving Sogroya® with the onset of a limp or complaints of persistent hip or knee pain for slipped capital femoral epiphysis and osteonecrosis and manage accordingly
- Progression of Preexisting Scoliosis in Pediatric Patients: Monitor patients with a history of scoliosis for disease progression
- Pancreatitis: Cases of pancreatitis have been reported in patients receiving somatropin. The risk may be greater in pediatric patients compared to adults. Consider pancreatitis in patients with persistent severe abdominal pain
- Lipohypertrophy/Lipoatrophy: May occur if Sogroya® is administered at the same site over a long period of time. Rotate injection sites to reduce this risk
- Sudden Death in Pediatric Patients with Prader-Willi Syndrome: There have been reports of fatalities after initiating therapy with somatropin in pediatric patients with Prader-Willi syndrome who had one or more of the following risk factors: severe obesity, history of upper airway obstruction or sleep apnea, or unidentified respiratory infection. Male patients with one or more of these factors may be at greater risk than females. Sogroya® is not indicated for the treatment of pediatric patients who have growth failure due to genetically confirmed Prader-Willi syndrome
- Laboratory Tests: Serum levels of inorganic phosphorus and alkaline phosphatase have increased after Sogroya® therapy. Serum levels of parathyroid hormone may increase with somatropin treatment
Adverse Reactions
- Common adverse reactions in pediatric patients treated with Sogroya® include: cough, diarrhea, ear infection, headache, injection site reaction, nasopharyngitis, pain in extremity, pyrexia, respiratory tract infection, and vomiting
- Adult patients with GHD: Adverse reactions reported in >2% of patients are: back pain, arthralgia, dyspepsia, sleep disorder, dizziness, tonsillitis, peripheral edema, vomiting, adrenal insufficiency, hypertension, blood creatine phosphokinase increase, weight increase, anemia
Drug Interactions
- Glucocorticoids: Patients treated with glucocorticoid for hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®
- Cytochrome P450-Metabolized Drugs: Sogroya® may alter the clearance. Monitor carefully if used with Sogroya®
- Oral Estrogen: Patients receiving oral estrogen replacement may require higher Sogroya® dosages
- Insulin and/or Other Antihyperglycemic Agents: Dose adjustment of insulin and/or antihyperglycemic agent may be required for patients with diabetes mellitus
Please click here for Sogroya® Prescribing Information.
Indications and Usage
Sogroya® (somapacitan-beco) injection 5 mg, 10 mg, or 15 mg is indicated for the:
- treatment of pediatric patients aged 2.5 years and older with:
- Growth failure due to inadequate secretion of endogenous growth hormone (GH)
- Short stature born Small for Gestational Age (SGA) and with no catch-up growth by 2 years of age
- Growth failure associated with Noonan Syndrome (NS)
- Idiopathic Short Stature (ISS)
- replacement of endogenous GH in adults with growth hormone deficiency (GHD)
Important Safety Information for Sogroya®
Contraindications
Sogroya® is contraindicated in patients with:
- acute critical illness after open-heart surgery, abdominal surgery, multiple accidental trauma, or acute respiratory failure because of the risk of increased mortality with use of Sogroya®
- hypersensitivity to Sogroya® or any of its excipients. Systemic hypersensitivity reactions have been reported postmarketing with somatropin
- pediatric patients with closed epiphyses
- active malignancy
- active proliferative or severe non-proliferative diabetic retinopathy
- pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to risk of sudden death
Important Safety Information for Sogroya®
Contraindications
Sogroya® is contraindicated in patients with:
- acute critical illness after open-heart surgery, abdominal surgery, multiple accidental trauma, or acute respiratory failure because of the risk of increased mortality with use of Sogroya®
- hypersensitivity to Sogroya® or any of its excipients. Systemic hypersensitivity reactions have been reported postmarketing with somatropin
- pediatric patients with closed epiphyses
- active malignancy
- active proliferative or severe non-proliferative diabetic retinopathy
- pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to risk of sudden death
Warnings & Precautions
- Increased Mortality in Patients with Acute Critical Illness: Increased mortality has been reported after treatment with somatropin in patients with acute critical illness due to complications following open-heart surgery, abdominal surgery, multiple accidental trauma, and in patients with acute respiratory failure
- Severe Hypersensitivity: Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported postmarketing with use of somatropin. Inform patients and/or caregivers that such reactions are possible and that prompt medical attention should be sought if an allergic reaction occurs
- Increased Risk of Neoplasms: There is an increased risk of malignancy progression with somatropin in patients with active malignancy. Any preexisting malignancy should be inactive, and its treatment complete prior to instituting Sogroya®. In childhood cancer survivors treated with radiation to the brain/head for their first neoplasm who developed subsequent GHD and were treated with somatropin, an increased risk of a second neoplasm has been reported. Monitor patients with a history of GHD secondary to an intracranial neoplasm for progression or recurrence of the tumor. Children with certain rare genetic causes of short stature have an increased risk of developing malignancies and should be carefully monitored for development of neoplasms. Monitor patients for increased growth or potential malignant changes of preexisting nevi. Advise patients/caregivers to report changes in the appearance of preexisting nevi
- Glucose Intolerance and Diabetes Mellitus: Treatment with somatropin may decrease insulin sensitivity, particularly at higher doses. New onset type 2 diabetes has been reported. Monitor glucose levels in all patients, especially in those with existing diabetes mellitus or with risk factors for diabetes mellitus, such as obesity, Turner syndrome, or a family history of diabetes mellitus. The doses of antidiabetic agents may require adjustment when Sogroya® is initiated
- Intracranial Hypertension: Has been reported usually within 8 weeks of treatment initiation. Perform fundoscopic examination prior to initiation of treatment and periodically thereafter. If papilledema is identified, evaluate the etiology, and treat the underlying cause before initiating Sogroya®. If papilledema is observed, stop treatment. If intracranial hypertension is confirmed, Sogroya® can be restarted at a lower dose after intracranial hypertension signs and symptoms have resolved
- Fluid Retention: Was observed during Sogroya® therapy. Clinical manifestations of fluid retention (e.g. edema and nerve compression syndromes, including carpal tunnel syndrome/paresthesia) are usually transient and dose dependent
- Hypoadrenalism: Patients receiving somatropin therapy who have or are at risk for corticotropin deficiency may be at risk for reduced serum cortisol levels and/or unmasking of central (secondary) hypoadrenalism. Patients treated with glucocorticoid replacement for previously diagnosed hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®. Monitor patients with known hypoadrenalism for reduced serum cortisol levels and/or need for glucocorticoid dose increases
- Hypothyroidism: Undiagnosed/untreated hypothyroidism may prevent an optimal response to Sogroya®. Monitor thyroid function periodically as hypothyroidism may occur or worsen after initiation of Sogroya®
- Slipped Capital Femoral Epiphysis in Pediatric Patients: Slipped capital femoral epiphysis may occur more frequently in patients undergoing rapid growth. Slipped capital femoral epiphysis may lead to osteonecrosis. Cases of slipped capital femoral epiphysis with or without osteonecrosis have been reported in pediatric patients with short stature receiving somatropin. Evaluate pediatric patients receiving Sogroya® with the onset of a limp or complaints of persistent hip or knee pain for slipped capital femoral epiphysis and osteonecrosis and manage accordingly
- Progression of Preexisting Scoliosis in Pediatric Patients: Monitor patients with a history of scoliosis for disease progression
- Pancreatitis: Cases of pancreatitis have been reported in patients receiving somatropin. The risk may be greater in pediatric patients compared to adults. Consider pancreatitis in patients with persistent severe abdominal pain
- Lipohypertrophy/Lipoatrophy: May occur if Sogroya® is administered at the same site over a long period of time. Rotate injection sites to reduce this risk
- Sudden Death in Pediatric Patients with Prader-Willi Syndrome: There have been reports of fatalities after initiating therapy with somatropin in pediatric patients with Prader-Willi syndrome who had one or more of the following risk factors: severe obesity, history of upper airway obstruction or sleep apnea, or unidentified respiratory infection. Male patients with one or more of these factors may be at greater risk than females. Sogroya® is not indicated for the treatment of pediatric patients who have growth failure due to genetically confirmed Prader-Willi syndrome
- Laboratory Tests: Serum levels of inorganic phosphorus and alkaline phosphatase have increased after Sogroya® therapy. Serum levels of parathyroid hormone may increase with somatropin treatment
Adverse Reactions
- Common adverse reactions in pediatric patients treated with Sogroya® include: cough, diarrhea, ear infection, headache, injection site reaction, nasopharyngitis, pain in extremity, pyrexia, respiratory tract infection, and vomiting
- Adult patients with GHD: Adverse reactions reported in >2% of patients are: back pain, arthralgia, dyspepsia, sleep disorder, dizziness, tonsillitis, peripheral edema, vomiting, adrenal insufficiency, hypertension, blood creatine phosphokinase increase, weight increase, anemia
Drug Interactions
- Glucocorticoids: Patients treated with glucocorticoid for hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®
- Cytochrome P450-Metabolized Drugs: Sogroya® may alter the clearance. Monitor carefully if used with Sogroya®
- Oral Estrogen: Patients receiving oral estrogen replacement may require higher Sogroya® dosages
- Insulin and/or Other Antihyperglycemic Agents: Dose adjustment of insulin and/or antihyperglycemic agent may be required for patients with diabetes mellitus
Please click here for Sogroya® Prescribing Information.
Important Safety Information for Sogroya®
Contraindications
Sogroya® is contraindicated in patients with:
- acute critical illness after open-heart surgery, abdominal surgery, multiple accidental trauma, or acute respiratory failure because of the risk of increased mortality with use of Sogroya®
- hypersensitivity to Sogroya® or any of its excipients. Systemic hypersensitivity reactions have been reported postmarketing with somatropin
- pediatric patients with closed epiphyses
- active malignancy
- active proliferative or severe non-proliferative diabetic retinopathy
- pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to risk of sudden death
Warnings & Precautions
- Increased Mortality in Patients with Acute Critical Illness: Increased mortality has been reported after treatment with somatropin in patients with acute critical illness due to complications following open-heart surgery, abdominal surgery, multiple accidental trauma, and in patients with acute respiratory failure
- Severe Hypersensitivity: Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported postmarketing with use of somatropin. Inform patients and/or caregivers that such reactions are possible and that prompt medical attention should be sought if an allergic reaction occurs
- Increased Risk of Neoplasms: There is an increased risk of malignancy progression with somatropin in patients with active malignancy. Any preexisting malignancy should be inactive, and its treatment complete prior to instituting Sogroya®. In childhood cancer survivors treated with radiation to the brain/head for their first neoplasm who developed subsequent GHD and were treated with somatropin, an increased risk of a second neoplasm has been reported. Monitor patients with a history of GHD secondary to an intracranial neoplasm for progression or recurrence of the tumor. Children with certain rare genetic causes of short stature have an increased risk of developing malignancies and should be carefully monitored for development of neoplasms. Monitor patients for increased growth or potential malignant changes of preexisting nevi. Advise patients/caregivers to report changes in the appearance of preexisting nevi
- Glucose Intolerance and Diabetes Mellitus: Treatment with somatropin may decrease insulin sensitivity, particularly at higher doses. New onset type 2 diabetes has been reported. Monitor glucose levels in all patients, especially in those with existing diabetes mellitus or with risk factors for diabetes mellitus, such as obesity, Turner syndrome, or a family history of diabetes mellitus. The doses of antidiabetic agents may require adjustment when Sogroya® is initiated
- Intracranial Hypertension: Has been reported usually within 8 weeks of treatment initiation. Perform fundoscopic examination prior to initiation of treatment and periodically thereafter. If papilledema is identified, evaluate the etiology, and treat the underlying cause before initiating Sogroya®. If papilledema is observed, stop treatment. If intracranial hypertension is confirmed, Sogroya® can be restarted at a lower dose after intracranial hypertension signs and symptoms have resolved
- Fluid Retention: Was observed during Sogroya® therapy. Clinical manifestations of fluid retention (e.g. edema and nerve compression syndromes, including carpal tunnel syndrome/paresthesia) are usually transient and dose dependent
- Hypoadrenalism: Patients receiving somatropin therapy who have or are at risk for corticotropin deficiency may be at risk for reduced serum cortisol levels and/or unmasking of central (secondary) hypoadrenalism. Patients treated with glucocorticoid replacement for previously diagnosed hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®. Monitor patients with known hypoadrenalism for reduced serum cortisol levels and/or need for glucocorticoid dose increases
- Hypothyroidism: Undiagnosed/untreated hypothyroidism may prevent an optimal response to Sogroya®. Monitor thyroid function periodically as hypothyroidism may occur or worsen after initiation of Sogroya®
- Slipped Capital Femoral Epiphysis in Pediatric Patients: Slipped capital femoral epiphysis may occur more frequently in patients undergoing rapid growth. Slipped capital femoral epiphysis may lead to osteonecrosis. Cases of slipped capital femoral epiphysis with or without osteonecrosis have been reported in pediatric patients with short stature receiving somatropin. Evaluate pediatric patients receiving Sogroya® with the onset of a limp or complaints of persistent hip or knee pain for slipped capital femoral epiphysis and osteonecrosis and manage accordingly
- Progression of Preexisting Scoliosis in Pediatric Patients: Monitor patients with a history of scoliosis for disease progression
- Pancreatitis: Cases of pancreatitis have been reported in patients receiving somatropin. The risk may be greater in pediatric patients compared to adults. Consider pancreatitis in patients with persistent severe abdominal pain
- Lipohypertrophy/Lipoatrophy: May occur if Sogroya® is administered at the same site over a long period of time. Rotate injection sites to reduce this risk
- Sudden Death in Pediatric Patients with Prader-Willi Syndrome: There have been reports of fatalities after initiating therapy with somatropin in pediatric patients with Prader-Willi syndrome who had one or more of the following risk factors: severe obesity, history of upper airway obstruction or sleep apnea, or unidentified respiratory infection. Male patients with one or more of these factors may be at greater risk than females. Sogroya® is not indicated for the treatment of pediatric patients who have growth failure due to genetically confirmed Prader-Willi syndrome
- Laboratory Tests: Serum levels of inorganic phosphorus and alkaline phosphatase have increased after Sogroya® therapy. Serum levels of parathyroid hormone may increase with somatropin treatment
Adverse Reactions
- Common adverse reactions in pediatric patients treated with Sogroya® include: cough, diarrhea, ear infection, headache, injection site reaction, nasopharyngitis, pain in extremity, pyrexia, respiratory tract infection, and vomiting
- Adult patients with GHD: Adverse reactions reported in >2% of patients are: back pain, arthralgia, dyspepsia, sleep disorder, dizziness, tonsillitis, peripheral edema, vomiting, adrenal insufficiency, hypertension, blood creatine phosphokinase increase, weight increase, anemia
Drug Interactions
- Glucocorticoids: Patients treated with glucocorticoid for hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®
- Cytochrome P450-Metabolized Drugs: Sogroya® may alter the clearance. Monitor carefully if used with Sogroya®
- Oral Estrogen: Patients receiving oral estrogen replacement may require higher Sogroya® dosages
- Insulin and/or Other Antihyperglycemic Agents: Dose adjustment of insulin and/or antihyperglycemic agent may be required for patients with diabetes mellitus
Please click here for Sogroya® Prescribing Information.
Indications and Usage
Sogroya® (somapacitan-beco) injection 5 mg, 10 mg, or 15 mg is indicated for the:
- treatment of pediatric patients aged 2.5 years and older with:
- Growth failure due to inadequate secretion of endogenous growth hormone (GH)
- Short stature born Small for Gestational Age (SGA) and with no catch-up growth by 2 years of age
- Growth failure associated with Noonan Syndrome (NS)
- Idiopathic Short Stature (ISS)
- replacement of endogenous GH in adults with growth hormone deficiency (GHD)
Indications and Usage
Sogroya® (somapacitan-beco) injection 5 mg, 10 mg, or 15 mg is indicated for the:
- treatment of pediatric patients aged 2.5 years and older with:
- Growth failure due to inadequate secretion of endogenous growth hormone (GH)
- Short stature born Small for Gestational Age (SGA) and with no catch-up growth by 2 years of age
- Growth failure associated with Noonan Syndrome (NS)
- Idiopathic Short Stature (ISS)
- replacement of endogenous GH in adults with growth hormone deficiency (GHD)
Important Safety Information for Sogroya®
Contraindications
Sogroya® is contraindicated in patients with:
- acute critical illness after open-heart surgery, abdominal surgery, multiple accidental trauma, or acute respiratory failure because of the risk of increased mortality with use of Sogroya®
- hypersensitivity to Sogroya® or any of its excipients. Systemic hypersensitivity reactions have been reported postmarketing with somatropin
- pediatric patients with closed epiphyses
- active malignancy
- active proliferative or severe non-proliferative diabetic retinopathy
- pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to risk of sudden death
Warnings & Precautions
- Increased Mortality in Patients with Acute Critical Illness: Increased mortality has been reported after treatment with somatropin in patients with acute critical illness due to complications following open-heart surgery, abdominal surgery, multiple accidental trauma, and in patients with acute respiratory failure
- Severe Hypersensitivity: Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported postmarketing with use of somatropin. Inform patients and/or caregivers that such reactions are possible and that prompt medical attention should be sought if an allergic reaction occurs
- Increased Risk of Neoplasms: There is an increased risk of malignancy progression with somatropin in patients with active malignancy. Any preexisting malignancy should be inactive, and its treatment complete prior to instituting Sogroya®. In childhood cancer survivors treated with radiation to the brain/head for their first neoplasm who developed subsequent GHD and were treated with somatropin, an increased risk of a second neoplasm has been reported. Monitor patients with a history of GHD secondary to an intracranial neoplasm for progression or recurrence of the tumor. Children with certain rare genetic causes of short stature have an increased risk of developing malignancies and should be carefully monitored for development of neoplasms. Monitor patients for increased growth or potential malignant changes of preexisting nevi. Advise patients/caregivers to report changes in the appearance of preexisting nevi
- Glucose Intolerance and Diabetes Mellitus: Treatment with somatropin may decrease insulin sensitivity, particularly at higher doses. New onset type 2 diabetes has been reported. Monitor glucose levels in all patients, especially in those with existing diabetes mellitus or with risk factors for diabetes mellitus, such as obesity, Turner syndrome, or a family history of diabetes mellitus. The doses of antidiabetic agents may require adjustment when Sogroya® is initiated
- Intracranial Hypertension: Has been reported usually within 8 weeks of treatment initiation. Perform fundoscopic examination prior to initiation of treatment and periodically thereafter. If papilledema is identified, evaluate the etiology, and treat the underlying cause before initiating Sogroya®. If papilledema is observed, stop treatment. If intracranial hypertension is confirmed, Sogroya® can be restarted at a lower dose after intracranial hypertension signs and symptoms have resolved
- Fluid Retention: Was observed during Sogroya® therapy. Clinical manifestations of fluid retention (e.g. edema and nerve compression syndromes, including carpal tunnel syndrome/paresthesia) are usually transient and dose dependent
- Hypoadrenalism: Patients receiving somatropin therapy who have or are at risk for corticotropin deficiency may be at risk for reduced serum cortisol levels and/or unmasking of central (secondary) hypoadrenalism. Patients treated with glucocorticoid replacement for previously diagnosed hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®. Monitor patients with known hypoadrenalism for reduced serum cortisol levels and/or need for glucocorticoid dose increases
- Hypothyroidism: Undiagnosed/untreated hypothyroidism may prevent an optimal response to Sogroya®. Monitor thyroid function periodically as hypothyroidism may occur or worsen after initiation of Sogroya®
- Slipped Capital Femoral Epiphysis in Pediatric Patients: Slipped capital femoral epiphysis may occur more frequently in patients undergoing rapid growth. Slipped capital femoral epiphysis may lead to osteonecrosis. Cases of slipped capital femoral epiphysis with or without osteonecrosis have been reported in pediatric patients with short stature receiving somatropin. Evaluate pediatric patients receiving Sogroya® with the onset of a limp or complaints of persistent hip or knee pain for slipped capital femoral epiphysis and osteonecrosis and manage accordingly
- Progression of Preexisting Scoliosis in Pediatric Patients: Monitor patients with a history of scoliosis for disease progression
- Pancreatitis: Cases of pancreatitis have been reported in patients receiving somatropin. The risk may be greater in pediatric patients compared to adults. Consider pancreatitis in patients with persistent severe abdominal pain
- Lipohypertrophy/Lipoatrophy: May occur if Sogroya® is administered at the same site over a long period of time. Rotate injection sites to reduce this risk
- Sudden Death in Pediatric Patients with Prader-Willi Syndrome: There have been reports of fatalities after initiating therapy with somatropin in pediatric patients with Prader-Willi syndrome who had one or more of the following risk factors: severe obesity, history of upper airway obstruction or sleep apnea, or unidentified respiratory infection. Male patients with one or more of these factors may be at greater risk than females. Sogroya® is not indicated for the treatment of pediatric patients who have growth failure due to genetically confirmed Prader-Willi syndrome
- Laboratory Tests: Serum levels of inorganic phosphorus and alkaline phosphatase have increased after Sogroya® therapy. Serum levels of parathyroid hormone may increase with somatropin treatment
Adverse Reactions
- Common adverse reactions in pediatric patients treated with Sogroya® include: cough, diarrhea, ear infection, headache, injection site reaction, nasopharyngitis, pain in extremity, pyrexia, respiratory tract infection, and vomiting
- Adult patients with GHD: Adverse reactions reported in >2% of patients are: back pain, arthralgia, dyspepsia, sleep disorder, dizziness, tonsillitis, peripheral edema, vomiting, adrenal insufficiency, hypertension, blood creatine phosphokinase increase, weight increase, anemia
Drug Interactions
- Glucocorticoids: Patients treated with glucocorticoid for hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®
- Cytochrome P450-Metabolized Drugs: Sogroya® may alter the clearance. Monitor carefully if used with Sogroya®
- Oral Estrogen: Patients receiving oral estrogen replacement may require higher Sogroya® dosages
- Insulin and/or Other Antihyperglycemic Agents: Dose adjustment of insulin and/or antihyperglycemic agent may be required for patients with diabetes mellitus
Please click here for Sogroya® Prescribing Information.
References:
- Data on file. Novo Nordisk, Inc; Plainsboro, NJ.
- Sogroya [package insert]. Plainsboro, NJ: Novo Nordisk, Inc.


