Sogroya® is indicated for the replacement of endogenous growth hormone in adults with growth hormone deficiency (GHD) and for the treatment of pediatric patients aged 2.5 years and older with: growth failure due to inadequate secretion of endogenous growth hormone (GH), short stature born Small for Gestational Age (SGA) and with no catch-up growth by 2 years of age, growth failure associated with Noonan Syndrome (NS), and Idiopathic Short Stature (ISS). Please see full indications.
WHY A ONCE-WEEKLY THERAPY?
WHY A ONCE-WEEKLY THERAPY?
Understand what obstacles may present for patients and caregivers
Understand how growth hormone deficiency (GHD) can affect pediatric and adult patients.
Missing multiple daily injections per week may substantially impact growth outcomes1
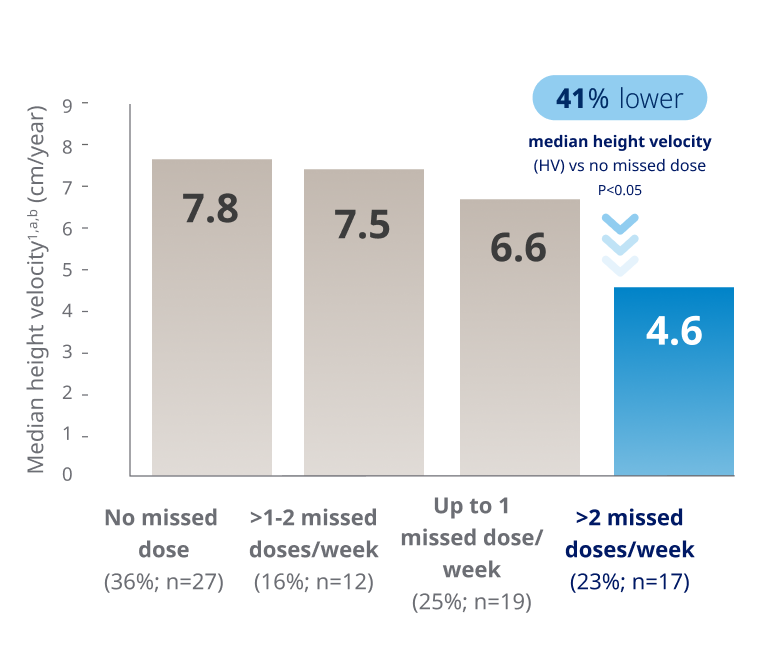
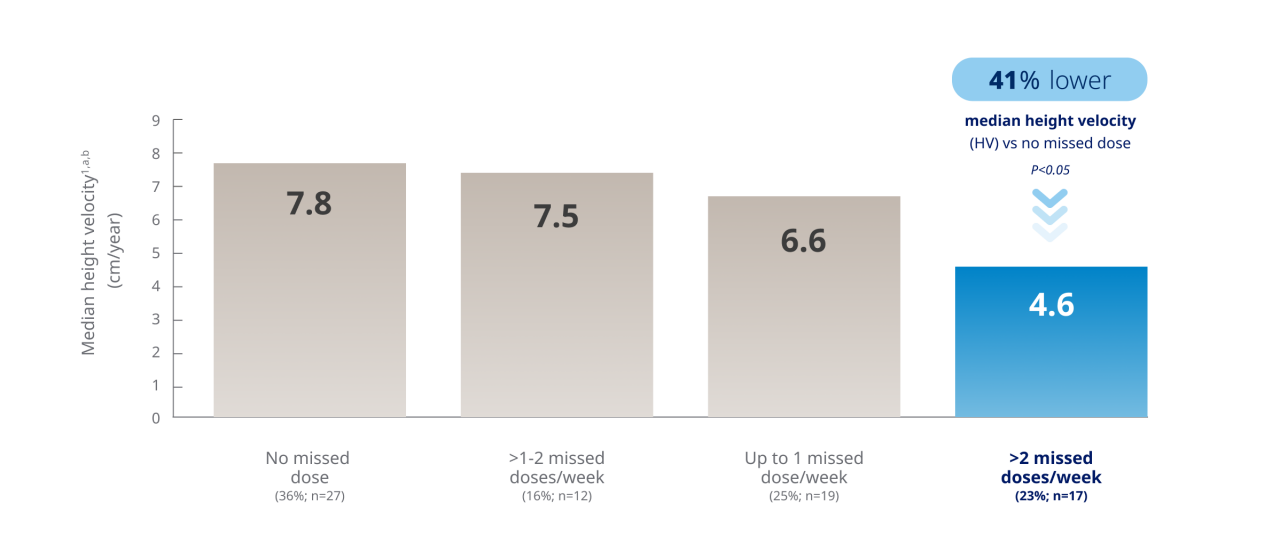
Chart based on Kapoor RR, et al. Monitoring of concordance in growth hormone therapy. Arch Dis Child. 2008;93(2):147-148.
Patients who missed >2 injections per week had a 41% lower median HV than those who missed none1,a,b
- Factors associated with lower adherence to therapy were a longer duration on GH therapy, lack of choice over GH device, and short duration (<4 weeks) of GH prescriptions
aUK study of 75 children with GHD who received GH therapy and attended a regional pediatric endocrine clinic from 1999 to 2003. 58 general practitioners (GPs) who issued GH prescriptions responded to a mailed questionnaire (66 GPs were contacted; 88% response rate) with the number of issued prescriptions and the total GH dose (or number of vials/cartridges) issued with each prescription during 3 specific 12-month periods (1999-2000, 2000-2001, and 2002-2003). Data on 11 patients were obtained from an outreach clinic where GH prescriptions were provided directly by a designated local consultant pediatrician. Adherence was assessed in each child by comparing total expected GH usage as documented in clinic records and letters to the total amount of GH prescribed by GPs during a 12-month period.1
bAdjusted for age and duration of GH therapy.1
GHD=growth hormone deficiency; HV=height velocity; GH=growth hormone.
In a systematic literature review and meta-analysis of medication adherence with once-weekly vs once-daily therapy,
Once-weekly treatment adherence was ~2X better than once-daily treatment2,c
cMeta-analysis of observational studies which were conducted in patients with osteoporosis.2
According to an LAGH Consensus Statement for pediatric GHD patients, developed independently by a panel of renowned pediatric endocrinology experts3:
- LAGH formulations offer a potential advantage over daily GH injections for children with GH deficiency in terms of reduced injection frequency and treatment burden; this may also be associated with improved adherence and treatment outcomes over time. However, long-term data is needed
- Parents/caregivers have shown a strong preference for the reduced frequency of LAGH preparations over daily GH formulations
- It is likely that most if not all children could be considered for a weekly administration regimen, particularly children or their caregivers who experience challenges with injections, as well as those at increased risk of non-adherence, frequent travel schedules, and split households
Consider if the flexibility of a once-weekly GH treatment option may impact your patient’s persistence and treatment experience
GH=growth hormone; GHD=growth hormone deficiency; LAGH=long-acting growth hormone.
Daily injections may present obstacles for patients and their caregivers
Patients with GHD continue to face challenges with daily injections4
Non-adherence to daily GH treatment is common and has been addressed in many studies5,6
Repeated injections are one of the main causes of non-adherence in pediatric-aged patients7

Children, adolescents, and their caregivers may struggle with continual daily injections over the long term4
Missed daily injections may impact growth outcomes8
GHD=growth hormone deficiency; GH=growth hormone
Missing
1 DAY
per week of daily
GH treatment
results in missing up to
52 DAYS
of treatment
in a year
which equates to missing
~1 YEAR
of doses over a 7-year
treatment period
GHD occurs in adults and has associated symptoms and conditions
Adult GHD (AGHD) is most often acquired from9,10:
A pituitary tumor9,10
Including treatment with surgery and/or radiation10
Trauma to the brain9
Other patient subpopulations include subarachnoid hemorrhage, ischemic stroke, and infections in the central nervous system9
AGHD may also be idiopathic or due to other organic causes.9,10
Symptoms and associated conditions of adult GHD include9:
Reduced energy levels
Reduced muscle strength
Osteoporosis
Lipid abnormalities
Increased total body fat
Insulin resistance
Impaired cardiac function
Symptoms and associated conditions of adult GHD include9:
Reduced energy levels
Reduced muscle strength
Osteoporosis
Lipid abnormalities
Increased total body fat
Insulin resistance
Impaired cardiac function
Management of adult GHD
Per the American Association of Clinical Endocrinologists (AACE), identifying adult patients with GHD includes assessing10:
PATIENTS WITH CLINICAL FEATURES SUGGESTIVE OF AGHD
eg, patients with organic hypothalamic-pituitary disease and low-serum IGF-1 levels
BIOCHEMICALLY PROVEN
EVIDENCE OF AGHD
eg, GH stimulation testing
Treatment options4,10
- With diagnosis confirmed, AACE recommends treating AGHD with growth hormone therapy10
- While once-daily GH treatment is considered the standard of care, according to GRS consensus, a once-weekly GH option may also help4
AGHD=adult growth hormone deficiency; GH=growth hormone; GHD=growth hormone deficiency; GRS=Growth Hormone Research Society; IGF-1=insulin-like growth factor-1.
How might treatment adherence affect pediatric patients with other specific growth-related conditions?
Early initiation and adherence to GH therapy may lead to improved growth outcomes in pediatric patients with ISS4,11-14
Treatment burden in ISS:

Diagnosis and treatment can be delayed, potentially reducing the treatment window15,16
- Average treatment duration is 4 to 7 years14

Treatment drop-out due to poor
adherence is common15
GH=growth hormone; ISS=Idiopathic Short Stature.
For children born SGA who are on GH therapy, treatment fatigue and poor adherence may lead to reduced growth outcomes17,18
Treatment burden in SGA:

In a cohort from an observational French registry study:
≈20% of patients (n=58/291) with SGA prematurely stopped GH treatment before reaching a normalized height17,d
Reasons for premature discontinuation included safety concerns, poor adherence, satisfaction with attained height, and patient, parent, or physician decision17
SGA can be associated with multiple comorbidities, which may increase treatment burden19,20

Parents of newborns born SGA may experience higher levels of psychological burden compared to those born normal for gestational age.21
dBased on an observational, multicenter, non-interventional French registry study that assessed the effectiveness and safety of daily GH in 1408 French children born SGA from 2005 to 2010. A cohort of 291 children were randomly selected for every fifth patient in the main study population to be followed for up to 6 months after the first visit and then annually until achieving final adult height or study termination. Final adult height was defined as height velocity <2 cm/year or bone age >14 years for girls and >16 years for boys. Normalized height was defined as height SDS >-2 from the reference population.
GH=growth hormone; SDS=standard deviation score; SGA=Small for Gestational Age.
Children with NS face a complex treatment burden, which may impact medication adherence22,23
Treatment burden in NS:
NS is a lifelong, multisystem condition with diverse complications and comorbidities that requires multidisciplinary care24,25

Late diagnosis may limit their GH treatment window22

Data suggest poor adherence to daily GH therapy may lead to a reduced height velocity for patients with NS23
GH=growth hormone; NS=Noonan Syndrome.


Missing 1 day per week of daily GH treatment is equivalent to missing 52 days of treatment in a year. If the treatment period is 7 years, this equates to skipping a full year of treatment.
Discover the pen
Discover the pen
Take a look at the features of the Sogroya® pen.
Take a look at the features of the Sogroya® pen.
How does it work?
How does it work?
Discover the science behind Sogroya®.
Discover the science behind Sogroya®.
Important Safety Information for Sogroya®
Contraindications
Sogroya® is contraindicated in patients with:
- acute critical illness after open-heart surgery, abdominal surgery, multiple accidental trauma, or acute respiratory failure because of the risk of increased mortality with use of Sogroya®
- hypersensitivity to Sogroya® or any of its excipients. Systemic hypersensitivity reactions have been reported postmarketing with somatropin
- pediatric patients with closed epiphyses
- active malignancy
- active proliferative or severe non-proliferative diabetic retinopathy
- pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to risk of sudden death
Warnings & Precautions
- Increased Mortality in Patients with Acute Critical Illness: Increased mortality has been reported after treatment with somatropin in patients with acute critical illness due to complications following open-heart surgery, abdominal surgery, multiple accidental trauma, and in patients with acute respiratory failure
- Severe Hypersensitivity: Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported postmarketing with use of somatropin. Inform patients and/or caregivers that such reactions are possible and that prompt medical attention should be sought if an allergic reaction occurs
- Increased Risk of Neoplasms: There is an increased risk of malignancy progression with somatropin in patients with active malignancy. Any preexisting malignancy should be inactive, and its treatment complete prior to instituting Sogroya®. In childhood cancer survivors treated with radiation to the brain/head for their first neoplasm who developed subsequent GHD and were treated with somatropin, an increased risk of a second neoplasm has been reported. Monitor patients with a history of GHD secondary to an intracranial neoplasm for progression or recurrence of the tumor. Children with certain rare genetic causes of short stature have an increased risk of developing malignancies and should be carefully monitored for development of neoplasms. Monitor patients for increased growth or potential malignant changes of preexisting nevi. Advise patients/caregivers to report changes in the appearance of preexisting nevi
- Glucose Intolerance and Diabetes Mellitus: Treatment with somatropin may decrease insulin sensitivity, particularly at higher doses. New onset type 2 diabetes has been reported. Monitor glucose levels in all patients, especially in those with existing diabetes mellitus or with risk factors for diabetes mellitus, such as obesity, Turner syndrome, or a family history of diabetes mellitus. The doses of antidiabetic agents may require adjustment when Sogroya® is initiated
- Intracranial Hypertension: Has been reported usually within 8 weeks of treatment initiation. Perform fundoscopic examination prior to initiation of treatment and periodically thereafter. If papilledema is identified, evaluate the etiology, and treat the underlying cause before initiating Sogroya®. If papilledema is observed, stop treatment. If intracranial hypertension is confirmed, Sogroya® can be restarted at a lower dose after intracranial hypertension signs and symptoms have resolved
- Fluid Retention: Was observed during Sogroya® therapy. Clinical manifestations of fluid retention (e.g. edema and nerve compression syndromes, including carpal tunnel syndrome/paresthesia) are usually transient and dose dependent
- Hypoadrenalism: Patients receiving somatropin therapy who have or are at risk for corticotropin deficiency may be at risk for reduced serum cortisol levels and/or unmasking of central (secondary) hypoadrenalism. Patients treated with glucocorticoid replacement for previously diagnosed hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®. Monitor patients with known hypoadrenalism for reduced serum cortisol levels and/or need for glucocorticoid dose increases
- Hypothyroidism: Undiagnosed/untreated hypothyroidism may prevent an optimal response to Sogroya®. Monitor thyroid function periodically as hypothyroidism may occur or worsen after initiation of Sogroya®
- Slipped Capital Femoral Epiphysis in Pediatric Patients: Slipped capital femoral epiphysis may occur more frequently in patients undergoing rapid growth. Slipped capital femoral epiphysis may lead to osteonecrosis. Cases of slipped capital femoral epiphysis with or without osteonecrosis have been reported in pediatric patients with short stature receiving somatropin. Evaluate pediatric patients receiving Sogroya® with the onset of a limp or complaints of persistent hip or knee pain for slipped capital femoral epiphysis and osteonecrosis and manage accordingly
- Progression of Preexisting Scoliosis in Pediatric Patients: Monitor patients with a history of scoliosis for disease progression
- Pancreatitis: Cases of pancreatitis have been reported in patients receiving somatropin. The risk may be greater in pediatric patients compared to adults. Consider pancreatitis in patients with persistent severe abdominal pain
- Lipohypertrophy/Lipoatrophy: May occur if Sogroya® is administered at the same site over a long period of time. Rotate injection sites to reduce this risk
- Sudden Death in Pediatric Patients with Prader-Willi Syndrome: There have been reports of fatalities after initiating therapy with somatropin in pediatric patients with Prader-Willi syndrome who had one or more of the following risk factors: severe obesity, history of upper airway obstruction or sleep apnea, or unidentified respiratory infection. Male patients with one or more of these factors may be at greater risk than females. Sogroya® is not indicated for the treatment of pediatric patients who have growth failure due to genetically confirmed Prader-Willi syndrome
- Laboratory Tests: Serum levels of inorganic phosphorus and alkaline phosphatase have increased after Sogroya® therapy. Serum levels of parathyroid hormone may increase with somatropin treatment
Adverse Reactions
- Common adverse reactions in pediatric patients treated with Sogroya® include: cough, diarrhea, ear infection, headache, injection site reaction, nasopharyngitis, pain in extremity, pyrexia, respiratory tract infection, and vomiting
- Adult patients with GHD: Adverse reactions reported in >2% of patients are: back pain, arthralgia, dyspepsia, sleep disorder, dizziness, tonsillitis, peripheral edema, vomiting, adrenal insufficiency, hypertension, blood creatine phosphokinase increase, weight increase, anemia
Drug Interactions
- Glucocorticoids: Patients treated with glucocorticoid for hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®
- Cytochrome P450-Metabolized Drugs: Sogroya® may alter the clearance. Monitor carefully if used with Sogroya®
- Oral Estrogen: Patients receiving oral estrogen replacement may require higher Sogroya® dosages
- Insulin and/or Other Antihyperglycemic Agents: Dose adjustment of insulin and/or antihyperglycemic agent may be required for patients with diabetes mellitus
Please click here for Sogroya® Prescribing Information.
Indications and Usage
Sogroya® (somapacitan-beco) injection 5 mg, 10 mg, or 15 mg is indicated for the:
- treatment of pediatric patients aged 2.5 years and older with:
- Growth failure due to inadequate secretion of endogenous growth hormone (GH)
- Short stature born Small for Gestational Age (SGA) and with no catch-up growth by 2 years of age
- Growth failure associated with Noonan Syndrome (NS)
- Idiopathic Short Stature (ISS)
- replacement of endogenous GH in adults with growth hormone deficiency (GHD)
Indications and Usage
Sogroya® (somapacitan-beco) injection 5 mg, 10 mg, or 15 mg is indicated for the:
- treatment of pediatric patients aged 2.5 years and older with:
- Growth failure due to inadequate secretion of endogenous growth hormone (GH)
- Short stature born Small for Gestational Age (SGA) and with no catch-up growth by 2 years of age
- Growth failure associated with Noonan Syndrome (NS)
- Idiopathic Short Stature (ISS)
- replacement of endogenous GH in adults with growth hormone deficiency (GHD)
Important Safety Information for Sogroya®
Contraindications
Sogroya® is contraindicated in patients with:
- acute critical illness after open-heart surgery, abdominal surgery, multiple accidental trauma, or acute respiratory failure because of the risk of increased mortality with use of Sogroya®
- hypersensitivity to Sogroya® or any of its excipients. Systemic hypersensitivity reactions have been reported postmarketing with somatropin
- pediatric patients with closed epiphyses
- active malignancy
- active proliferative or severe non-proliferative diabetic retinopathy
- pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to risk of sudden death
Warnings & Precautions
- Increased Mortality in Patients with Acute Critical Illness: Increased mortality has been reported after treatment with somatropin in patients with acute critical illness due to complications following open-heart surgery, abdominal surgery, multiple accidental trauma, and in patients with acute respiratory failure
- Severe Hypersensitivity: Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported postmarketing with use of somatropin. Inform patients and/or caregivers that such reactions are possible and that prompt medical attention should be sought if an allergic reaction occurs
- Increased Risk of Neoplasms: There is an increased risk of malignancy progression with somatropin in patients with active malignancy. Any preexisting malignancy should be inactive, and its treatment complete prior to instituting Sogroya®. In childhood cancer survivors treated with radiation to the brain/head for their first neoplasm who developed subsequent GHD and were treated with somatropin, an increased risk of a second neoplasm has been reported. Monitor patients with a history of GHD secondary to an intracranial neoplasm for progression or recurrence of the tumor. Children with certain rare genetic causes of short stature have an increased risk of developing malignancies and should be carefully monitored for development of neoplasms. Monitor patients for increased growth or potential malignant changes of preexisting nevi. Advise patients/caregivers to report changes in the appearance of preexisting nevi
- Glucose Intolerance and Diabetes Mellitus: Treatment with somatropin may decrease insulin sensitivity, particularly at higher doses. New onset type 2 diabetes has been reported. Monitor glucose levels in all patients, especially in those with existing diabetes mellitus or with risk factors for diabetes mellitus, such as obesity, Turner syndrome, or a family history of diabetes mellitus. The doses of antidiabetic agents may require adjustment when Sogroya® is initiated
- Intracranial Hypertension: Has been reported usually within 8 weeks of treatment initiation. Perform fundoscopic examination prior to initiation of treatment and periodically thereafter. If papilledema is identified, evaluate the etiology, and treat the underlying cause before initiating Sogroya®. If papilledema is observed, stop treatment. If intracranial hypertension is confirmed, Sogroya® can be restarted at a lower dose after intracranial hypertension signs and symptoms have resolved
- Fluid Retention: Was observed during Sogroya® therapy. Clinical manifestations of fluid retention (e.g. edema and nerve compression syndromes, including carpal tunnel syndrome/paresthesia) are usually transient and dose dependent
- Hypoadrenalism: Patients receiving somatropin therapy who have or are at risk for corticotropin deficiency may be at risk for reduced serum cortisol levels and/or unmasking of central (secondary) hypoadrenalism. Patients treated with glucocorticoid replacement for previously diagnosed hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®. Monitor patients with known hypoadrenalism for reduced serum cortisol levels and/or need for glucocorticoid dose increases
- Hypothyroidism: Undiagnosed/untreated hypothyroidism may prevent an optimal response to Sogroya®. Monitor thyroid function periodically as hypothyroidism may occur or worsen after initiation of Sogroya®
- Slipped Capital Femoral Epiphysis in Pediatric Patients: Slipped capital femoral epiphysis may occur more frequently in patients undergoing rapid growth. Slipped capital femoral epiphysis may lead to osteonecrosis. Cases of slipped capital femoral epiphysis with or without osteonecrosis have been reported in pediatric patients with short stature receiving somatropin. Evaluate pediatric patients receiving Sogroya® with the onset of a limp or complaints of persistent hip or knee pain for slipped capital femoral epiphysis and osteonecrosis and manage accordingly
- Progression of Preexisting Scoliosis in Pediatric Patients: Monitor patients with a history of scoliosis for disease progression
- Pancreatitis: Cases of pancreatitis have been reported in patients receiving somatropin. The risk may be greater in pediatric patients compared to adults. Consider pancreatitis in patients with persistent severe abdominal pain
- Lipohypertrophy/Lipoatrophy: May occur if Sogroya® is administered at the same site over a long period of time. Rotate injection sites to reduce this risk
- Sudden Death in Pediatric Patients with Prader-Willi Syndrome: There have been reports of fatalities after initiating therapy with somatropin in pediatric patients with Prader-Willi syndrome who had one or more of the following risk factors: severe obesity, history of upper airway obstruction or sleep apnea, or unidentified respiratory infection. Male patients with one or more of these factors may be at greater risk than females. Sogroya® is not indicated for the treatment of pediatric patients who have growth failure due to genetically confirmed Prader-Willi syndrome
- Laboratory Tests: Serum levels of inorganic phosphorus and alkaline phosphatase have increased after Sogroya® therapy. Serum levels of parathyroid hormone may increase with somatropin treatment
Adverse Reactions
- Common adverse reactions in pediatric patients treated with Sogroya® include: cough, diarrhea, ear infection, headache, injection site reaction, nasopharyngitis, pain in extremity, pyrexia, respiratory tract infection, and vomiting
- Adult patients with GHD: Adverse reactions reported in >2% of patients are: back pain, arthralgia, dyspepsia, sleep disorder, dizziness, tonsillitis, peripheral edema, vomiting, adrenal insufficiency, hypertension, blood creatine phosphokinase increase, weight increase, anemia
Drug Interactions
- Glucocorticoids: Patients treated with glucocorticoid for hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®
- Cytochrome P450-Metabolized Drugs: Sogroya® may alter the clearance. Monitor carefully if used with Sogroya®
- Oral Estrogen: Patients receiving oral estrogen replacement may require higher Sogroya® dosages
- Insulin and/or Other Antihyperglycemic Agents: Dose adjustment of insulin and/or antihyperglycemic agent may be required for patients with diabetes mellitus
Please click here for Sogroya® Prescribing Information.
Indications and Usage
Sogroya® (somapacitan-beco) injection 5 mg, 10 mg, or 15 mg is indicated for the:
- treatment of pediatric patients aged 2.5 years and older with:
- Growth failure due to inadequate secretion of endogenous growth hormone (GH)
- Short stature born Small for Gestational Age (SGA) and with no catch-up growth by 2 years of age
- Growth failure associated with Noonan Syndrome (NS)
- Idiopathic Short Stature (ISS)
- replacement of endogenous GH in adults with growth hormone deficiency (GHD)
Important Safety Information for Sogroya®
Contraindications
Sogroya® is contraindicated in patients with:
- acute critical illness after open-heart surgery, abdominal surgery, multiple accidental trauma, or acute respiratory failure because of the risk of increased mortality with use of Sogroya®
- hypersensitivity to Sogroya® or any of its excipients. Systemic hypersensitivity reactions have been reported postmarketing with somatropin
- pediatric patients with closed epiphyses
- active malignancy
- active proliferative or severe non-proliferative diabetic retinopathy
- pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to risk of sudden death
Important Safety Information for Sogroya®
Contraindications
Sogroya® is contraindicated in patients with:
- acute critical illness after open-heart surgery, abdominal surgery, multiple accidental trauma, or acute respiratory failure because of the risk of increased mortality with use of Sogroya®
- hypersensitivity to Sogroya® or any of its excipients. Systemic hypersensitivity reactions have been reported postmarketing with somatropin
- pediatric patients with closed epiphyses
- active malignancy
- active proliferative or severe non-proliferative diabetic retinopathy
- pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to risk of sudden death
Warnings & Precautions
- Increased Mortality in Patients with Acute Critical Illness: Increased mortality has been reported after treatment with somatropin in patients with acute critical illness due to complications following open-heart surgery, abdominal surgery, multiple accidental trauma, and in patients with acute respiratory failure
- Severe Hypersensitivity: Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported postmarketing with use of somatropin. Inform patients and/or caregivers that such reactions are possible and that prompt medical attention should be sought if an allergic reaction occurs
- Increased Risk of Neoplasms: There is an increased risk of malignancy progression with somatropin in patients with active malignancy. Any preexisting malignancy should be inactive, and its treatment complete prior to instituting Sogroya®. In childhood cancer survivors treated with radiation to the brain/head for their first neoplasm who developed subsequent GHD and were treated with somatropin, an increased risk of a second neoplasm has been reported. Monitor patients with a history of GHD secondary to an intracranial neoplasm for progression or recurrence of the tumor. Children with certain rare genetic causes of short stature have an increased risk of developing malignancies and should be carefully monitored for development of neoplasms. Monitor patients for increased growth or potential malignant changes of preexisting nevi. Advise patients/caregivers to report changes in the appearance of preexisting nevi
- Glucose Intolerance and Diabetes Mellitus: Treatment with somatropin may decrease insulin sensitivity, particularly at higher doses. New onset type 2 diabetes has been reported. Monitor glucose levels in all patients, especially in those with existing diabetes mellitus or with risk factors for diabetes mellitus, such as obesity, Turner syndrome, or a family history of diabetes mellitus. The doses of antidiabetic agents may require adjustment when Sogroya® is initiated
- Intracranial Hypertension: Has been reported usually within 8 weeks of treatment initiation. Perform fundoscopic examination prior to initiation of treatment and periodically thereafter. If papilledema is identified, evaluate the etiology, and treat the underlying cause before initiating Sogroya®. If papilledema is observed, stop treatment. If intracranial hypertension is confirmed, Sogroya® can be restarted at a lower dose after intracranial hypertension signs and symptoms have resolved
- Fluid Retention: Was observed during Sogroya® therapy. Clinical manifestations of fluid retention (e.g. edema and nerve compression syndromes, including carpal tunnel syndrome/paresthesia) are usually transient and dose dependent
- Hypoadrenalism: Patients receiving somatropin therapy who have or are at risk for corticotropin deficiency may be at risk for reduced serum cortisol levels and/or unmasking of central (secondary) hypoadrenalism. Patients treated with glucocorticoid replacement for previously diagnosed hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®. Monitor patients with known hypoadrenalism for reduced serum cortisol levels and/or need for glucocorticoid dose increases
- Hypothyroidism: Undiagnosed/untreated hypothyroidism may prevent an optimal response to Sogroya®. Monitor thyroid function periodically as hypothyroidism may occur or worsen after initiation of Sogroya®
- Slipped Capital Femoral Epiphysis in Pediatric Patients: Slipped capital femoral epiphysis may occur more frequently in patients undergoing rapid growth. Slipped capital femoral epiphysis may lead to osteonecrosis. Cases of slipped capital femoral epiphysis with or without osteonecrosis have been reported in pediatric patients with short stature receiving somatropin. Evaluate pediatric patients receiving Sogroya® with the onset of a limp or complaints of persistent hip or knee pain for slipped capital femoral epiphysis and osteonecrosis and manage accordingly
- Progression of Preexisting Scoliosis in Pediatric Patients: Monitor patients with a history of scoliosis for disease progression
- Pancreatitis: Cases of pancreatitis have been reported in patients receiving somatropin. The risk may be greater in pediatric patients compared to adults. Consider pancreatitis in patients with persistent severe abdominal pain
- Lipohypertrophy/Lipoatrophy: May occur if Sogroya® is administered at the same site over a long period of time. Rotate injection sites to reduce this risk
- Sudden Death in Pediatric Patients with Prader-Willi Syndrome: There have been reports of fatalities after initiating therapy with somatropin in pediatric patients with Prader-Willi syndrome who had one or more of the following risk factors: severe obesity, history of upper airway obstruction or sleep apnea, or unidentified respiratory infection. Male patients with one or more of these factors may be at greater risk than females. Sogroya® is not indicated for the treatment of pediatric patients who have growth failure due to genetically confirmed Prader-Willi syndrome
- Laboratory Tests: Serum levels of inorganic phosphorus and alkaline phosphatase have increased after Sogroya® therapy. Serum levels of parathyroid hormone may increase with somatropin treatment
Adverse Reactions
- Common adverse reactions in pediatric patients treated with Sogroya® include: cough, diarrhea, ear infection, headache, injection site reaction, nasopharyngitis, pain in extremity, pyrexia, respiratory tract infection, and vomiting
- Adult patients with GHD: Adverse reactions reported in >2% of patients are: back pain, arthralgia, dyspepsia, sleep disorder, dizziness, tonsillitis, peripheral edema, vomiting, adrenal insufficiency, hypertension, blood creatine phosphokinase increase, weight increase, anemia
Drug Interactions
- Glucocorticoids: Patients treated with glucocorticoid for hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®
- Cytochrome P450-Metabolized Drugs: Sogroya® may alter the clearance. Monitor carefully if used with Sogroya®
- Oral Estrogen: Patients receiving oral estrogen replacement may require higher Sogroya® dosages
- Insulin and/or Other Antihyperglycemic Agents: Dose adjustment of insulin and/or antihyperglycemic agent may be required for patients with diabetes mellitus
Please click here for Sogroya® Prescribing Information.
Important Safety Information for Sogroya®
Contraindications
Sogroya® is contraindicated in patients with:
- acute critical illness after open-heart surgery, abdominal surgery, multiple accidental trauma, or acute respiratory failure because of the risk of increased mortality with use of Sogroya®
- hypersensitivity to Sogroya® or any of its excipients. Systemic hypersensitivity reactions have been reported postmarketing with somatropin
- pediatric patients with closed epiphyses
- active malignancy
- active proliferative or severe non-proliferative diabetic retinopathy
- pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to risk of sudden death
Warnings & Precautions
- Increased Mortality in Patients with Acute Critical Illness: Increased mortality has been reported after treatment with somatropin in patients with acute critical illness due to complications following open-heart surgery, abdominal surgery, multiple accidental trauma, and in patients with acute respiratory failure
- Severe Hypersensitivity: Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported postmarketing with use of somatropin. Inform patients and/or caregivers that such reactions are possible and that prompt medical attention should be sought if an allergic reaction occurs
- Increased Risk of Neoplasms: There is an increased risk of malignancy progression with somatropin in patients with active malignancy. Any preexisting malignancy should be inactive, and its treatment complete prior to instituting Sogroya®. In childhood cancer survivors treated with radiation to the brain/head for their first neoplasm who developed subsequent GHD and were treated with somatropin, an increased risk of a second neoplasm has been reported. Monitor patients with a history of GHD secondary to an intracranial neoplasm for progression or recurrence of the tumor. Children with certain rare genetic causes of short stature have an increased risk of developing malignancies and should be carefully monitored for development of neoplasms. Monitor patients for increased growth or potential malignant changes of preexisting nevi. Advise patients/caregivers to report changes in the appearance of preexisting nevi
- Glucose Intolerance and Diabetes Mellitus: Treatment with somatropin may decrease insulin sensitivity, particularly at higher doses. New onset type 2 diabetes has been reported. Monitor glucose levels in all patients, especially in those with existing diabetes mellitus or with risk factors for diabetes mellitus, such as obesity, Turner syndrome, or a family history of diabetes mellitus. The doses of antidiabetic agents may require adjustment when Sogroya® is initiated
- Intracranial Hypertension: Has been reported usually within 8 weeks of treatment initiation. Perform fundoscopic examination prior to initiation of treatment and periodically thereafter. If papilledema is identified, evaluate the etiology, and treat the underlying cause before initiating Sogroya®. If papilledema is observed, stop treatment. If intracranial hypertension is confirmed, Sogroya® can be restarted at a lower dose after intracranial hypertension signs and symptoms have resolved
- Fluid Retention: Was observed during Sogroya® therapy. Clinical manifestations of fluid retention (e.g. edema and nerve compression syndromes, including carpal tunnel syndrome/paresthesia) are usually transient and dose dependent
- Hypoadrenalism: Patients receiving somatropin therapy who have or are at risk for corticotropin deficiency may be at risk for reduced serum cortisol levels and/or unmasking of central (secondary) hypoadrenalism. Patients treated with glucocorticoid replacement for previously diagnosed hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®. Monitor patients with known hypoadrenalism for reduced serum cortisol levels and/or need for glucocorticoid dose increases
- Hypothyroidism: Undiagnosed/untreated hypothyroidism may prevent an optimal response to Sogroya®. Monitor thyroid function periodically as hypothyroidism may occur or worsen after initiation of Sogroya®
- Slipped Capital Femoral Epiphysis in Pediatric Patients: Slipped capital femoral epiphysis may occur more frequently in patients undergoing rapid growth. Slipped capital femoral epiphysis may lead to osteonecrosis. Cases of slipped capital femoral epiphysis with or without osteonecrosis have been reported in pediatric patients with short stature receiving somatropin. Evaluate pediatric patients receiving Sogroya® with the onset of a limp or complaints of persistent hip or knee pain for slipped capital femoral epiphysis and osteonecrosis and manage accordingly
- Progression of Preexisting Scoliosis in Pediatric Patients: Monitor patients with a history of scoliosis for disease progression
- Pancreatitis: Cases of pancreatitis have been reported in patients receiving somatropin. The risk may be greater in pediatric patients compared to adults. Consider pancreatitis in patients with persistent severe abdominal pain
- Lipohypertrophy/Lipoatrophy: May occur if Sogroya® is administered at the same site over a long period of time. Rotate injection sites to reduce this risk
- Sudden Death in Pediatric Patients with Prader-Willi Syndrome: There have been reports of fatalities after initiating therapy with somatropin in pediatric patients with Prader-Willi syndrome who had one or more of the following risk factors: severe obesity, history of upper airway obstruction or sleep apnea, or unidentified respiratory infection. Male patients with one or more of these factors may be at greater risk than females. Sogroya® is not indicated for the treatment of pediatric patients who have growth failure due to genetically confirmed Prader-Willi syndrome
- Laboratory Tests: Serum levels of inorganic phosphorus and alkaline phosphatase have increased after Sogroya® therapy. Serum levels of parathyroid hormone may increase with somatropin treatment
Adverse Reactions
- Common adverse reactions in pediatric patients treated with Sogroya® include: cough, diarrhea, ear infection, headache, injection site reaction, nasopharyngitis, pain in extremity, pyrexia, respiratory tract infection, and vomiting
- Adult patients with GHD: Adverse reactions reported in >2% of patients are: back pain, arthralgia, dyspepsia, sleep disorder, dizziness, tonsillitis, peripheral edema, vomiting, adrenal insufficiency, hypertension, blood creatine phosphokinase increase, weight increase, anemia
Drug Interactions
- Glucocorticoids: Patients treated with glucocorticoid for hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®
- Cytochrome P450-Metabolized Drugs: Sogroya® may alter the clearance. Monitor carefully if used with Sogroya®
- Oral Estrogen: Patients receiving oral estrogen replacement may require higher Sogroya® dosages
- Insulin and/or Other Antihyperglycemic Agents: Dose adjustment of insulin and/or antihyperglycemic agent may be required for patients with diabetes mellitus
Please click here for Sogroya® Prescribing Information.
Indications and Usage
Sogroya® (somapacitan-beco) injection 5 mg, 10 mg, or 15 mg is indicated for the:
- treatment of pediatric patients aged 2.5 years and older with:
- Growth failure due to inadequate secretion of endogenous growth hormone (GH)
- Short stature born Small for Gestational Age (SGA) and with no catch-up growth by 2 years of age
- Growth failure associated with Noonan Syndrome (NS)
- Idiopathic Short Stature (ISS)
- replacement of endogenous GH in adults with growth hormone deficiency (GHD)
Indications and Usage
Sogroya® (somapacitan-beco) injection 5 mg, 10 mg, or 15 mg is indicated for the:
- treatment of pediatric patients aged 2.5 years and older with:
- Growth failure due to inadequate secretion of endogenous growth hormone (GH)
- Short stature born Small for Gestational Age (SGA) and with no catch-up growth by 2 years of age
- Growth failure associated with Noonan Syndrome (NS)
- Idiopathic Short Stature (ISS)
- replacement of endogenous GH in adults with growth hormone deficiency (GHD)
Important Safety Information for Sogroya®
Contraindications
Sogroya® is contraindicated in patients with:
- acute critical illness after open-heart surgery, abdominal surgery, multiple accidental trauma, or acute respiratory failure because of the risk of increased mortality with use of Sogroya®
- hypersensitivity to Sogroya® or any of its excipients. Systemic hypersensitivity reactions have been reported postmarketing with somatropin
- pediatric patients with closed epiphyses
- active malignancy
- active proliferative or severe non-proliferative diabetic retinopathy
- pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to risk of sudden death
Warnings & Precautions
- Increased Mortality in Patients with Acute Critical Illness: Increased mortality has been reported after treatment with somatropin in patients with acute critical illness due to complications following open-heart surgery, abdominal surgery, multiple accidental trauma, and in patients with acute respiratory failure
- Severe Hypersensitivity: Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported postmarketing with use of somatropin. Inform patients and/or caregivers that such reactions are possible and that prompt medical attention should be sought if an allergic reaction occurs
- Increased Risk of Neoplasms: There is an increased risk of malignancy progression with somatropin in patients with active malignancy. Any preexisting malignancy should be inactive, and its treatment complete prior to instituting Sogroya®. In childhood cancer survivors treated with radiation to the brain/head for their first neoplasm who developed subsequent GHD and were treated with somatropin, an increased risk of a second neoplasm has been reported. Monitor patients with a history of GHD secondary to an intracranial neoplasm for progression or recurrence of the tumor. Children with certain rare genetic causes of short stature have an increased risk of developing malignancies and should be carefully monitored for development of neoplasms. Monitor patients for increased growth or potential malignant changes of preexisting nevi. Advise patients/caregivers to report changes in the appearance of preexisting nevi
- Glucose Intolerance and Diabetes Mellitus: Treatment with somatropin may decrease insulin sensitivity, particularly at higher doses. New onset type 2 diabetes has been reported. Monitor glucose levels in all patients, especially in those with existing diabetes mellitus or with risk factors for diabetes mellitus, such as obesity, Turner syndrome, or a family history of diabetes mellitus. The doses of antidiabetic agents may require adjustment when Sogroya® is initiated
- Intracranial Hypertension: Has been reported usually within 8 weeks of treatment initiation. Perform fundoscopic examination prior to initiation of treatment and periodically thereafter. If papilledema is identified, evaluate the etiology, and treat the underlying cause before initiating Sogroya®. If papilledema is observed, stop treatment. If intracranial hypertension is confirmed, Sogroya® can be restarted at a lower dose after intracranial hypertension signs and symptoms have resolved
- Fluid Retention: Was observed during Sogroya® therapy. Clinical manifestations of fluid retention (e.g. edema and nerve compression syndromes, including carpal tunnel syndrome/paresthesia) are usually transient and dose dependent
- Hypoadrenalism: Patients receiving somatropin therapy who have or are at risk for corticotropin deficiency may be at risk for reduced serum cortisol levels and/or unmasking of central (secondary) hypoadrenalism. Patients treated with glucocorticoid replacement for previously diagnosed hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®. Monitor patients with known hypoadrenalism for reduced serum cortisol levels and/or need for glucocorticoid dose increases
- Hypothyroidism: Undiagnosed/untreated hypothyroidism may prevent an optimal response to Sogroya®. Monitor thyroid function periodically as hypothyroidism may occur or worsen after initiation of Sogroya®
- Slipped Capital Femoral Epiphysis in Pediatric Patients: Slipped capital femoral epiphysis may occur more frequently in patients undergoing rapid growth. Slipped capital femoral epiphysis may lead to osteonecrosis. Cases of slipped capital femoral epiphysis with or without osteonecrosis have been reported in pediatric patients with short stature receiving somatropin. Evaluate pediatric patients receiving Sogroya® with the onset of a limp or complaints of persistent hip or knee pain for slipped capital femoral epiphysis and osteonecrosis and manage accordingly
- Progression of Preexisting Scoliosis in Pediatric Patients: Monitor patients with a history of scoliosis for disease progression
- Pancreatitis: Cases of pancreatitis have been reported in patients receiving somatropin. The risk may be greater in pediatric patients compared to adults. Consider pancreatitis in patients with persistent severe abdominal pain
- Lipohypertrophy/Lipoatrophy: May occur if Sogroya® is administered at the same site over a long period of time. Rotate injection sites to reduce this risk
- Sudden Death in Pediatric Patients with Prader-Willi Syndrome: There have been reports of fatalities after initiating therapy with somatropin in pediatric patients with Prader-Willi syndrome who had one or more of the following risk factors: severe obesity, history of upper airway obstruction or sleep apnea, or unidentified respiratory infection. Male patients with one or more of these factors may be at greater risk than females. Sogroya® is not indicated for the treatment of pediatric patients who have growth failure due to genetically confirmed Prader-Willi syndrome
- Laboratory Tests: Serum levels of inorganic phosphorus and alkaline phosphatase have increased after Sogroya® therapy. Serum levels of parathyroid hormone may increase with somatropin treatment
Adverse Reactions
- Common adverse reactions in pediatric patients treated with Sogroya® include: cough, diarrhea, ear infection, headache, injection site reaction, nasopharyngitis, pain in extremity, pyrexia, respiratory tract infection, and vomiting
- Adult patients with GHD: Adverse reactions reported in >2% of patients are: back pain, arthralgia, dyspepsia, sleep disorder, dizziness, tonsillitis, peripheral edema, vomiting, adrenal insufficiency, hypertension, blood creatine phosphokinase increase, weight increase, anemia
Drug Interactions
- Glucocorticoids: Patients treated with glucocorticoid for hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Sogroya®
- Cytochrome P450-Metabolized Drugs: Sogroya® may alter the clearance. Monitor carefully if used with Sogroya®
- Oral Estrogen: Patients receiving oral estrogen replacement may require higher Sogroya® dosages
- Insulin and/or Other Antihyperglycemic Agents: Dose adjustment of insulin and/or antihyperglycemic agent may be required for patients with diabetes mellitus
Please click here for Sogroya® Prescribing Information.
References:
- Kapoor RR, Burke SA, Sparrow SE, et al. Monitoring of concordance in growth hormone therapy. Arch Dis Child.2008;93(2):147-148. doi:10.1136/adc.2006.114249
- Iglay K, Cao X, Mavros P, Joshi K, Yu S, Tuncell K. Systematic literature review and meta-analysis of medication adherence with once-weekly versus once-daily therapy. Clin Ther. 2015;37(8):1813-1821.e1. doi:10.1016/j.clinthera.2015.05.505
- Maniatis A, Cutfield W, Dattani M, et al. Long-acting growth hormone therapy in pediatric growth hormone deficiency: a consensus statement. J Clin Endocrinol Metab. Published online December 3, 2024. doi:10.1210/clinem/dgae834
- Christiansen JS, Backeljauw PF, Bidlingmaier M, et al. Growth Hormone Research Society perspective on the development of long-acting growth hormone preparations. Eur J Endocrinol. 2016;174(6):C1–C8. doi:10.1530/EJE-16-0111
- Graham S, Weinman J, Auyeung V. Identifying potentially modifiable factors associated with treatment non-adherence in paediatric growth hormone deficiency: a systematic review. Horm Res Paediatr. 2018;90(4):221-227. doi:10.1159/000493211
- Bagnasco F, Di Iorgi N, Roveda A, Gallizia A, Haupt R, Maghnie M; Adherence Investigators Group. Prevalence and correlates of adherence in children and adolescents treated with growth hormone: a multicenter Italian study. Endocr Pract. 2017;23(8):929-941. doi:10.4158/EP171786.OR
- Mameli C, Orso M, Calcaterra V, et al. Efficacy, safety, quality of life, adherence and cost-effectiveness of long-acting growth hormone replacement therapy compared to daily growth hormone in children with growth hormone deficiency: a systematic review and meta-analysis. Pharmacol Res. 2023;193:106805. doi:10.1016/j.phrs/2023.106805
- Loftus J, Chen Y, Alvir JMJ, et al. Suboptimal adherence to daily growth hormone in a US real-world study: an unmet need in the treatment of pediatric growth hormone deficiency. Curr Med Res Opin. 2021;37(12):2141-2150. doi: 10.1080/03007995.2021.1982682
- National Organization for Rare Disorders (NORD). Growth hormone deficiency. Updated 2016. Accessed November 17, 2022. https://rarediseases.org/rare-diseases/growth-hormone- deficiency
- Yuen KCJ, Beverly MKB, Radovick S, et al. American Association of Clinical Endocrinologists and American College of Endocrinology guidelines for management of growth hormone deficiency in adults and patients transitioning from pediatric to adult care. Endocr Pract. 2019;25(11):1191-1232.
- Ranke MB. Treatment of children and adolescents with idiopathic short stature. Nat Rev Endocrinol. 2013;9(6):325-334. doi: 10.1038/nrendo.2013.71
- Polak M, Blair J, Kotnik P, Pournara E, Pedersen BT, Rohrer TR. Early growth hormone treatment start in childhood growth hormone deficiency improves near adult height: analysis from NordiNet® International Outcome Study. Eur J Endocrinol. 2017;177(5):421-429. doi:10.1530/EJE-16-1024
- Pedicelli S, Peschiaroli E, Violi E, Cianfarani S. Controversies in the definition and treatment of idiopathic short stature (ISS). J Clin Res Pediatr Endocrinol. 2009;1(3):105-115. doi:10.4008/jcrpe.v1i3.53
- Cohen P, Rogol AD, Deal CL, et al. Consensus statement on the diagnosis and treatment of children with idiopathic short stature: a summary of the Growth Hormone Research Society, the Lawson Wilkins Pediatric Endocrine Society, and the European Society for Paediatric Endocrinology Workshop. J Clin Endocrinol Metab. 2008;93(11):4210-4217. doi: 10.1210/jc.2008-0509
- Wu W, Li N, Wang T, Luo X. Real-life growth hormone treatment patterns in children from China: a report from two databases. Adv Ther. 2025;42(7):3562-3575. doi:10.1007/s12325-025-03204-9
- Ben-Ari T, Chodick G, Shalev V, Goldstein D, Gomez R, Landau Z. Real-world treatment patterns and outcomes of growth hormone treatment among children in Israel over the past decade (2004- 2015). Front Pediatr. 2021;9:711979. doi: 10.3389/fped.2021.711979
- Salles J-P, Coutant R, Leheup B, Nicolina M, Hacques E, Villette B. Small for gestational age patients with premature treatment discontinuation: their journey in French, real-life settings. Poster presented at: 57th Meeting of the European Society for Paediatric Endocrinology; September 27-29, 2018; Athens, Greece. Poster P3-P200.
- De Pedro S, Murillo M, Salinas I, et al. Variability in adherence to rhGH treatment: Socioeconomic causes and effect on children's growth. Growth Harm IGF Res. 2015;25(6):280-283. doi:10.1016/j.ghir.2015.12.002
- Ferrigno R, Savanelli MC, Cioffi D, Pellino V, Klain A. Auxological and metabolic effects of long-term treatment with recombinant growth hormone in children born small for gestational age: a retrospective study. Endocrine. 2024;84(1):213-222. doi:10.1007/s12020-023-03665-4
- Jung H, Rosilio M, Blum WF, Drop SL. Growth hormone treatment for short stature in children born small for gestational age. Adv Ther. 2008;25(10):951-978. doi:10.1007/s12325-008-0101-3
- Bellido-González M, Robles-Ortega H, Castelar-Ríos MJ, e t al. Psychological distress and resilience of mothers and fathers with respect to the neurobehavioral performance of small-for-gestational-age newborns. Health Qual Life Outcomes. 2019;17(1):54. doi:10.1186/s12955-019-1119-8
- Seo GH, Yoo HW. Growth hormone therapy in patients with Noonan syndrome. Ann Pediatr Endocrinol Metab. 2018;23(4):176-181. doi:10.6065/apem.2018.23.4.176
- Kappelgaard AM, Laursen T. The benefits of growth hormone therapy in patients with Turner syndrome, Noonan syndrome and children born small for gestational age. Growth Hormone & IGF Research. 2011;21 (6):253-262.
- Stagi S, Cappa M, Gagliardi MG, Tartaglia M, Maghnie M, Italian Multidisciplinary Noonan Syndrome Working Group. Multidisciplinary Treatment of Patients With Noonan Syndrome: A Consensus Statement. JAMA Netw Open.2025;8(10):e2537603. doi:10.1001 /jamanetworkopen.2025.37603
- Rohrer TR, Abuzzahab J, Backeljauw P, et al. Long-term effectiveness and safety of childhood growth hormone treatment in Noonan syndrome. Horm Res Paediatr. 2020.