Safety profiles for once-weekly Ozempic® (semaglutide) injection and once-daily Ozempic® pill
Safety and tolerability evaluated across clinical trials
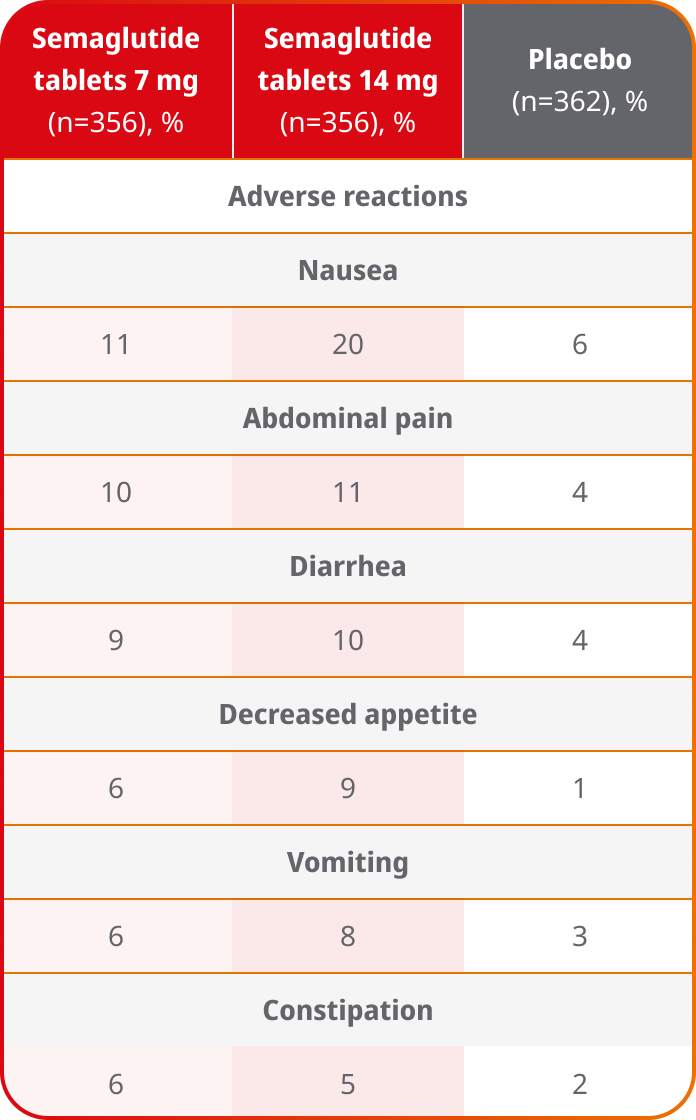
Rates of GI symptoms across placebo-controlled trials with Ozempic® injection and semaglutide tablets1,2,a
- The most common adverse reactions reported in ≥5% of patients taking Ozempic® injection and Ozempic® tablets are nausea, vomiting, diarrhea, abdominal pain, and constipation. Decreased appetite was also reported in ≥5% of patients taking semaglutide tablets
- The majority of reports of nausea, vomiting, and/or diarrhea occurred during dose escalation
- Ozempic® injection: In placebo-controlled trials, discontinuation due to GI adverse reactions was 3.1% and 3.8% for the Ozempic® injection 0.5 mg and 1 mg dose, respectively, compared with 0.4% for placebo
- Semaglutide tablets: 4% and 8% of patients discontinued semaglutide tablets 7 mg and 14 mg, respectively, due to GI adverse reactions, compared with 1% of patients receiving placebo
Rates of hypoglycemia in placebo-controlled trials1-3
- Incidence of severe hypoglycemia was <1.5% across placebo-controlled trials with Ozempic® injection, (<1% for Ozempic® 1 mg and Ozempic® 2 mg in SUSTAIN FORTE), and <1% in placebo-controlled trials with semaglutide tablets1,2,a-c
- Patients receiving Ozempic® injection and semaglutide tablets, used in combination with insulin secretagogues (eg, sulfonylureas) or insulin, may have an increased risk of hypoglycemia, including severe hypoglycemia1,2
- When initiating Ozempic® injection and semaglutide tablets, patients may require a lower dose of the insulin secretagogue (such as sulfonylurea) or insulin to reduce the risk of hypoglycemia1-3
No new safety signals identified with Ozempic® injection 2 mg dose1,3
- In a 40-week, randomized, active-controlled trial with 959 patients treated with Ozempic® injection 1 mg or Ozempic® injection 2 mg once weekly as add-on to metformin with or without sulfonylurea treatment for 40 weeks, no new safety signals were identified1
- In the trial with Ozempic® injection 1 mg and 2 mg, GI adverse reactions occurred more frequently among patients receiving Ozempic® injection 2 mg (34.0%) vs Ozempic® injection 1 mg (30.8%)1
- SUSTAIN FORTE: Results are from a 40-week, randomized, active-controlled trial in 959 adult patients with type 2 diabetes on metformin with or without a sulfonylurea, comparing Ozempic® injection 1 mg with 2 mg1,3
There were no new serious or severe adverse reactions identified in the FLOW trial1
- In the FLOW trial, in patients with type 2 diabetes and chronic kidney disease treated with Ozempic® injection, safety data collection was limited to serious adverse events and selected predefined categories of adverse events regardless of seriousness1
Ozempic® tablets 1.5 mg, 4 mg, and 9 mg are an updated formulation with increased bioavailability. This updated formulation is established with comparable mean exposure and peak concentration. The safety and effectiveness of Ozempic® tablets were established through adequate and well-controlled studies of semaglutide tablets 3 mg, 7 mg, and 14 mg.2,4
aIncluding 1 monotherapy trial and 1 trial in combination with insulin. The medications in parentheses represent background medications in each trial. PIONEER 7, 9, and 10 were included as part of the safety analysis.1,3
bIn addition to the CVOT (SUSTAIN 6), glycemic control trials (SUSTAIN 1 through 5), and SUSTAIN FORTE, 2 Japanese trials are incorporated into the pool of safety data within the FDA-approved label. These trials evaluated the use of Ozempic® as monotherapy and add-on therapy to oral medications or insulin. CV safety was assessed in SUSTAIN 6. Occurrence of adverse reactions was evaluated in SUSTAIN 1-5 and 2 Japanese trials.1,3
cSevere hypoglycemia adverse reactions are episodes requiring the assistance of another person.1,3
AEs occurring in ≥5% of participants treated with Ozempic® injection in SUSTAIN 45
SUSTAIN 4 was not designed to evaluate relative safety between Ozempic® injection and Lantus®
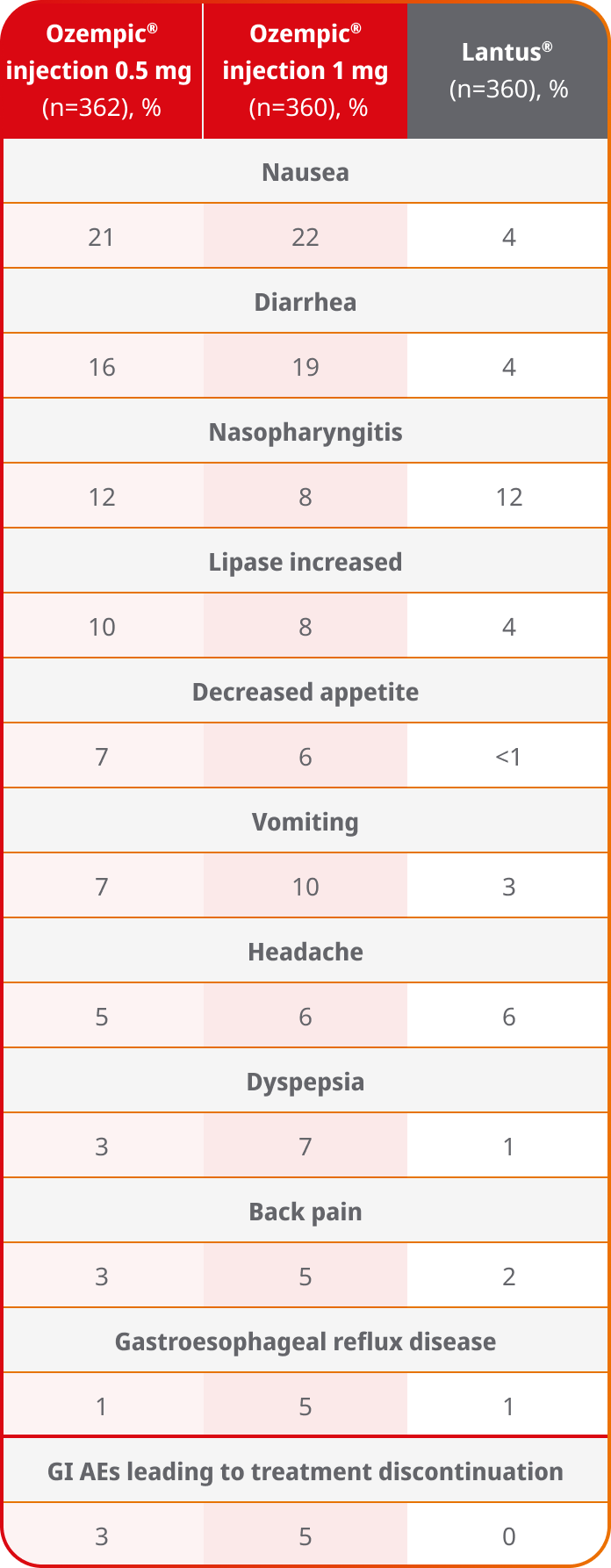

- In placebo-controlled trials, the most common adverse reactions reported in ≥5% of patients treated with Ozempic® injection are nausea, vomiting, diarrhea, abdominal pain, and constipation1
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice1
- Comparator adverse event rates are not an adequate basis for comparison of safety between products1
- Incidence of severe hypoglycemia was ≤1.5% across all placebo-controlled trials1
- Incidence of severe hypoglycemia or blood glucose-confirmed hypoglycemia (% of patients) was 4% with Ozempic® injection 0.5 mg, 6% with Ozempic® injection 1 mg, and 11% with Lantus®5,a
Results are from a 30-week, randomized, open-label, active-controlled trial in 1082 adult, insulin-naive patients with type 2 diabetes on metformin ± sulfonylurea.1,5
aDefined as an event requiring assistance of another person to actively administer carbohydrates, glucagon, or take other corrective actions or blood glucose–confirmed symptomatic hypoglycemia (plasma glucose ≤3.1 mmol/L [56 mg/dL]).5
AE=adverse event; GI=gastrointestinal.
AEs occurring in ≥5% of participants treated with Ozempic® injection in SUSTAIN 56,7

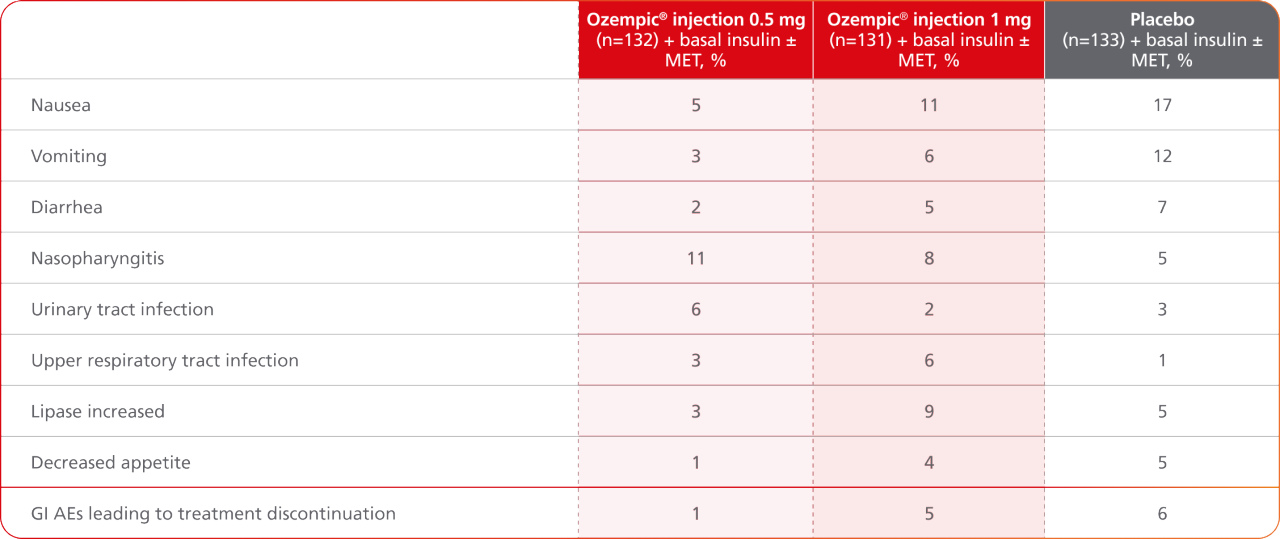
- Incidence of severe hypoglycemia was ≤1.5% across all placebo-controlled trials1
- Incidence of severe hypoglycemia or blood glucose-confirmed hypoglycemia (% of patients) was6:
- 8.3% with Ozempic® injection 0.5 mg + basal insulin ± MET
- 10.7% with Ozempic® injection 1 mg + basal insulin ± MET
- 5.3% with placebo + basal insulin ± MET
- Patients receiving Ozempic® injection in combination with an insulin secretagogue (eg, sulfonylurea) or insulin may have an increased risk of hypoglycemia, including severe hypoglycemia1
Results are from a 30-week, randomized, double-blind, placebo-controlled, parallel-group trial in 397 adult patients with type 2 diabetes evaluating the addition of Ozempic® injection 0.5 mg and Ozempic® injection 1 mg to basal insulin ± MET.1
AE=adverse events; GI=gastrointestinal; MET=metformin.
AEs occurring in ≥5% of participants treated with Ozempic® injection in SUSTAIN 68
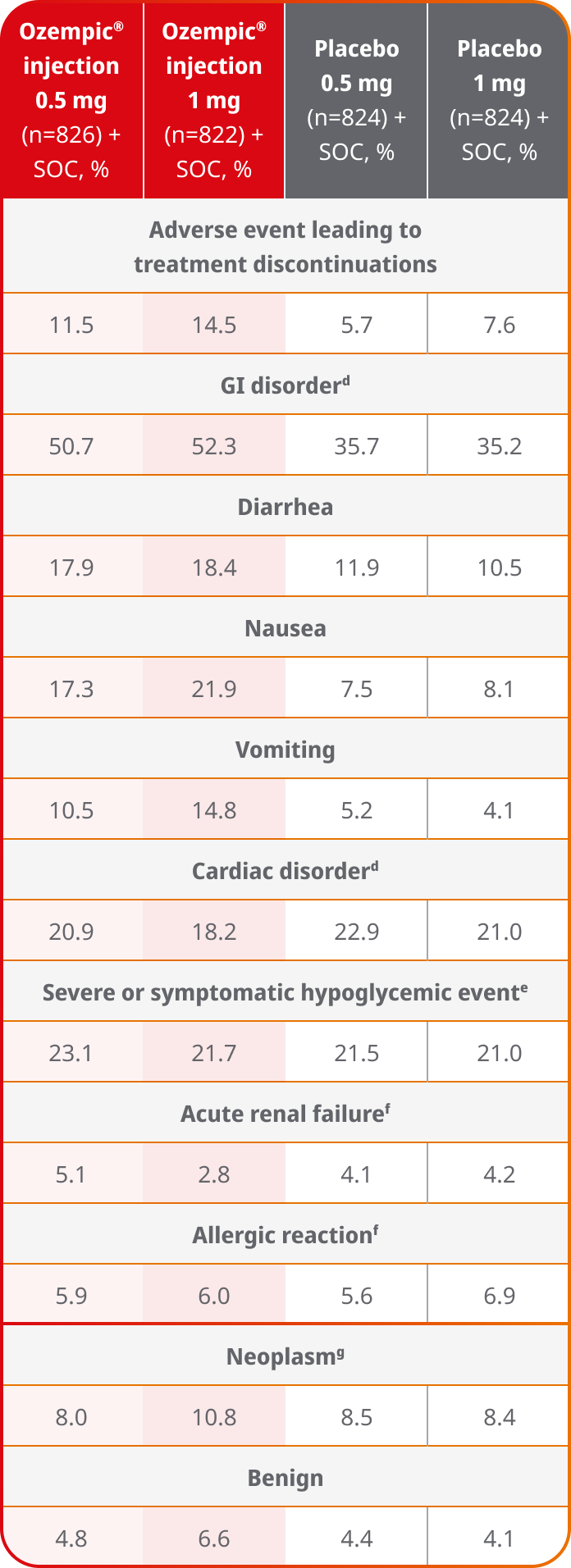
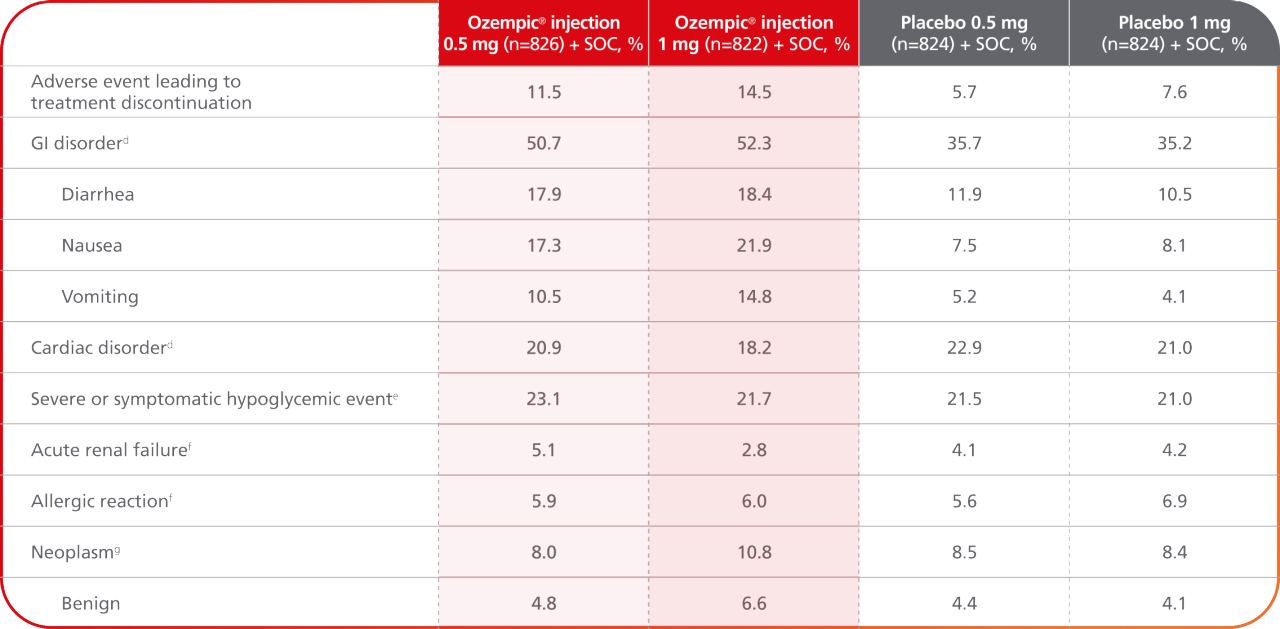
- Treatment discontinuation due to adverse events was more frequent in the Ozempic® injection group than in the placebo group8
- GI disorders were more frequent in the Ozempic® injection group than in the placebo group8
dThis category was defined according to the system organ class in MedDRA.8
eThis category of hypoglycemic event includes episodes of severe hypoglycemia (defined according to the American Diabetes Association criteria) or symptomatic hypoglycemia as confirmed on plasma glucose testing (<56 mg/dL [3.1 mmoL/l]).8
fThis category was based on the group of preferred terms in MedDRA.8
gThis event was confirmed by the event adjudication committee.8
AE=adverse event; GI=gastrointestinal; MedDRA=Medical Dictionary for Regulatory Activities; SOC=standard of care.
AEs occurring in ≥5% of participants treated with Ozempic® injection in SUSTAIN 79
SUSTAIN 7 was not designed to evaluate relative safety between Ozempic® injection and Trulicity®
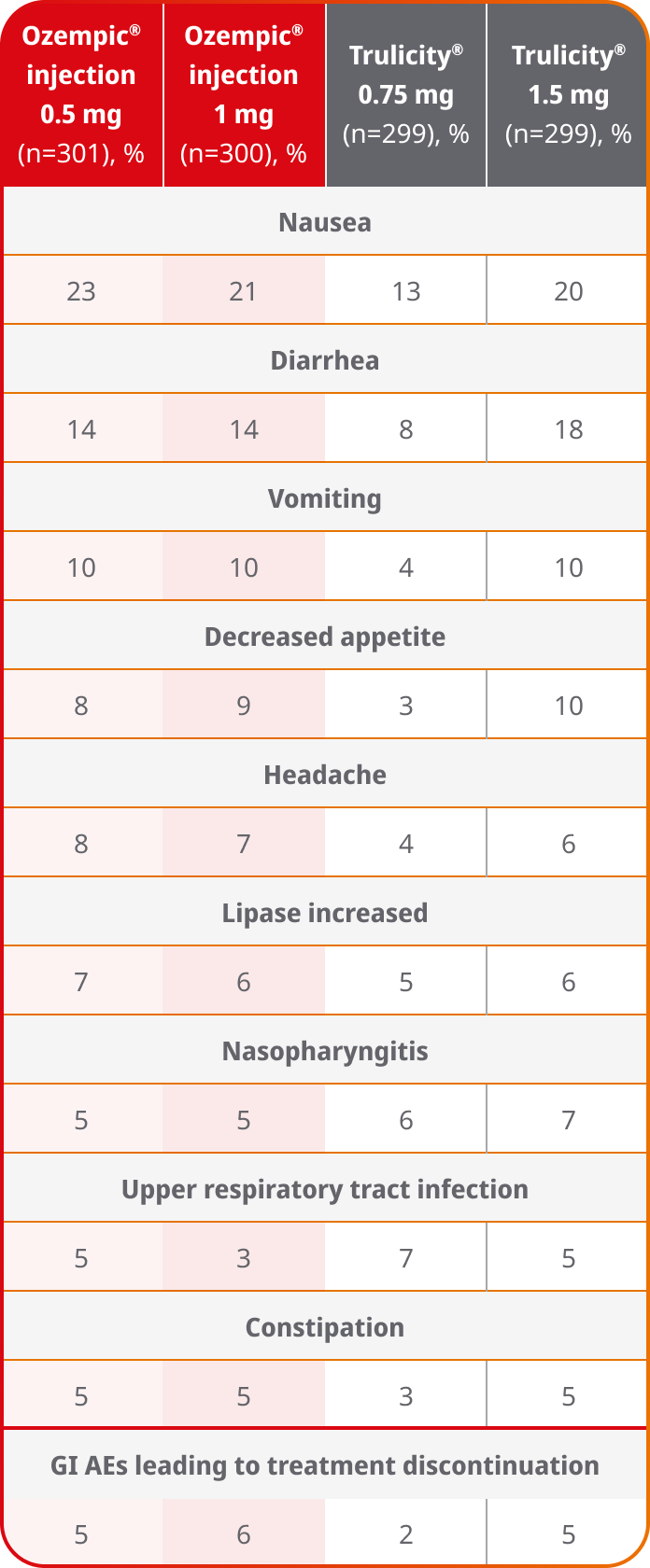
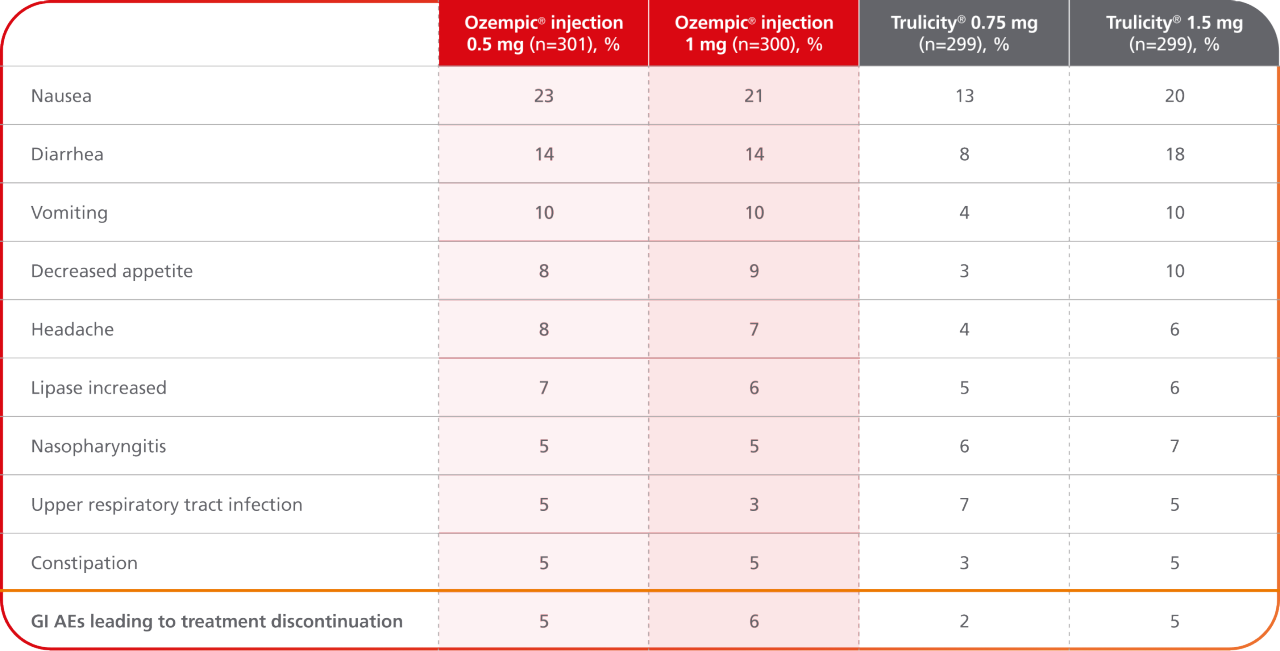
- In placebo-controlled trials, the most common adverse reactions reported in ≥5% of patients treated with Ozempic® injection are nausea, vomiting, diarrhea, abdominal pain, and constipation1
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice1
- Comparator adverse event rates are not an adequate basis for comparison of safety between products9
Results are from a 40-week, randomized, open-label, active-controlled trial in 1201 adult patients with type 2 diabetes on metformin comparing Ozempic® injection 0.5 mg with Trulicity® 0.75 mg and Ozempic® injection 1 mg with Trulicity® 1.5 mg.3
AE=adverse event; GI=gastrointestinal.
AEs occurring in ≥5% of participants treated with Ozempic® injection 1 mg and 2 mg in SUSTAIN FORTE3
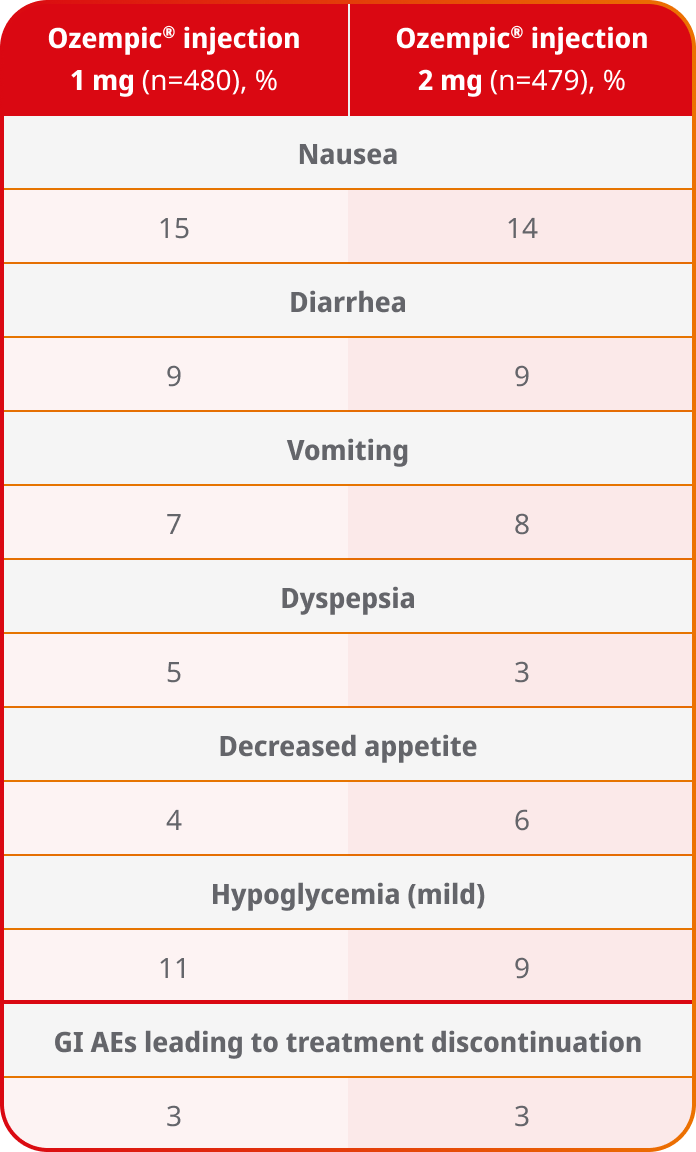

- In placebo-controlled trials, the most common adverse reactions reported in ≥5% of patients treated with Ozempic® injection are nausea, vomiting, diarrhea, abdominal pain, and constipation1
- No new safety signals were identified. GI adverse reactions occurred more frequently among patients receiving Ozempic® injection 2 mg (34.0%) vs Ozempic® injection 1 mg (30.8%)1
- Incidence of severe hypoglycemia (Level 3) was less than 1% for Ozempic® injection 1 mg and Ozempic® injection 2 mg in the SUSTAIN FORTE trial3
Safety results are from a 40-week, randomized, active-controlled, parallel-group trial in 961 adult patients with type 2 diabetes in need of treatment intensification comparing Ozempic® injection 2 mg with Ozempic® injection 1 mg.1,3
AE=adverse event; GI=gastrointestinal.
Prespecified adverse events of special interest10
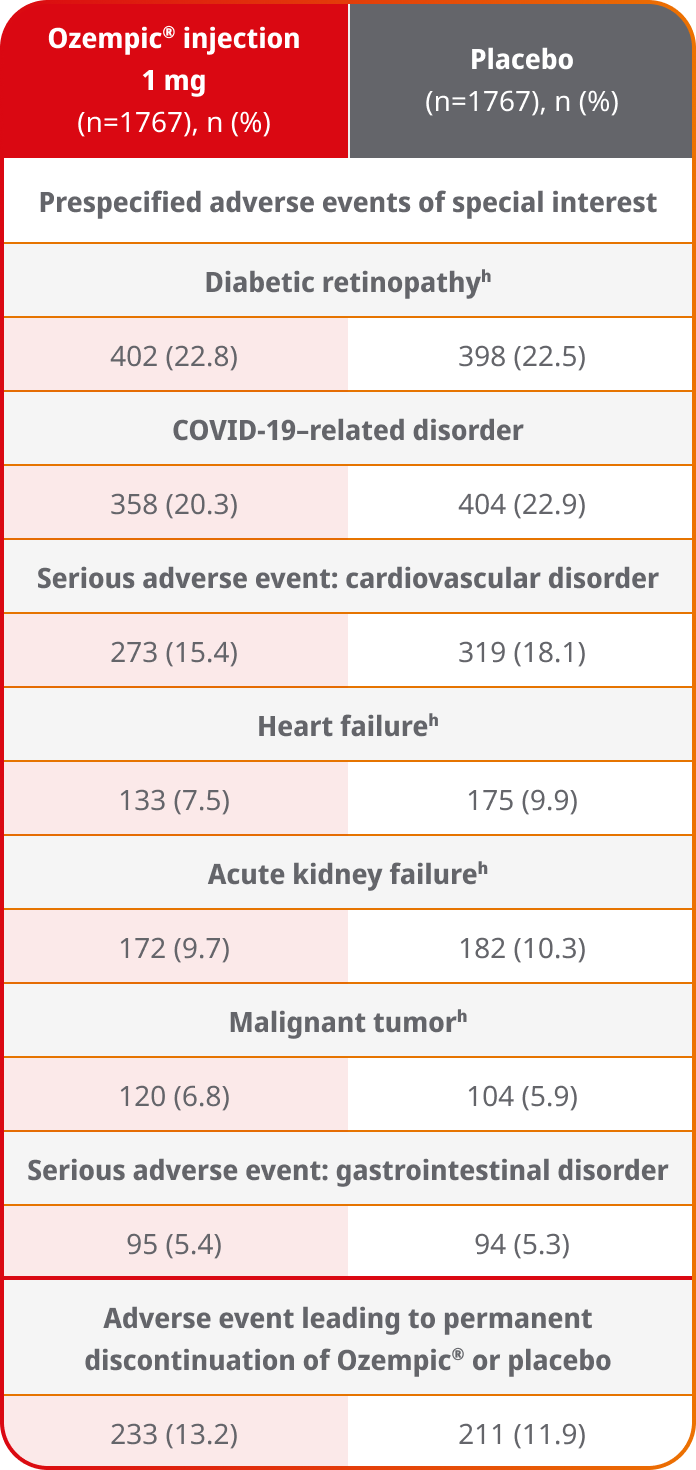
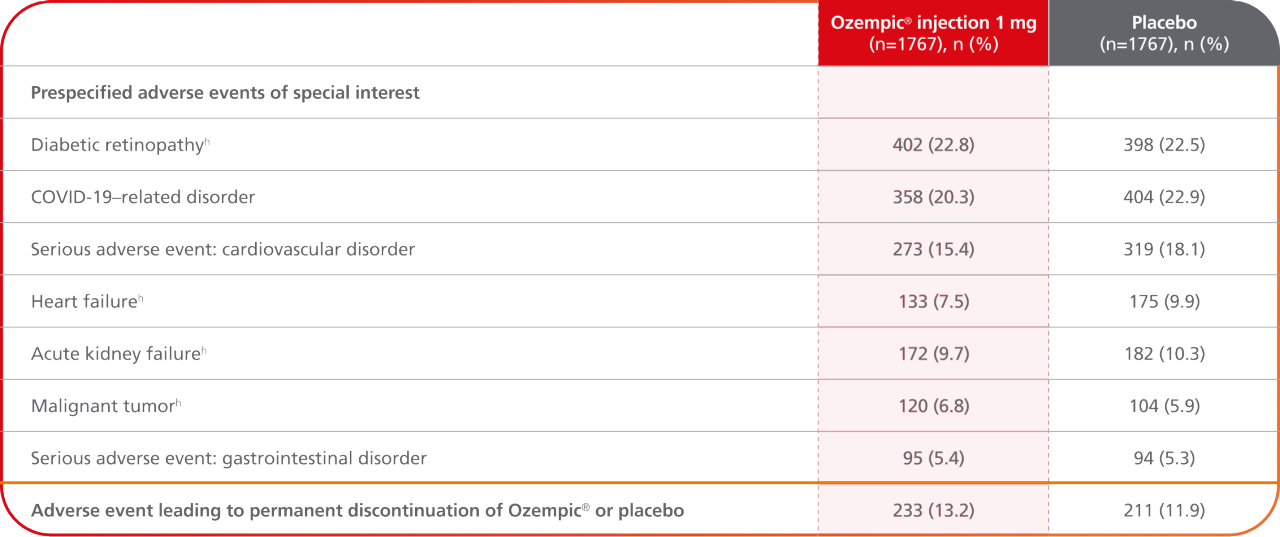
- In FLOW, the most common adverse event of special interest was diabetic retinopathy10
- The incidence of serious adverse events related to a cardiovascular disorder was approximately 15% with Ozempic® injection and 18% with placebo10
- In the FLOW trial, safety data collection was limited to serious adverse events and selected predefined categories of adverse events regardless of seriousness. There were no new serious or severe adverse reactions identified in this trial1
hData were from an additional data-collection form; data for all other prespecified events of special interest were collected by means of a Medical Dictionary for Regulatory Activities search.10

The data provided on the Rybelsus® PIONEER Clinical Trial program are based on clinical trials involving Rybelsus® (3 mg, 7 mg, and 14 mg). The efficacy and safety of Ozempic® pill (1.5 mg, 4 mg, and 9 mg) have been established based on adequate and well-controlled studies of Rybelsus® (3 mg, 7 mg, and 14 mg).2
In the pool of placebo- and active-controlled trials, the types and frequency of common adverse reactions, excluding hypoglycemia, were similar to those listed in the table above.
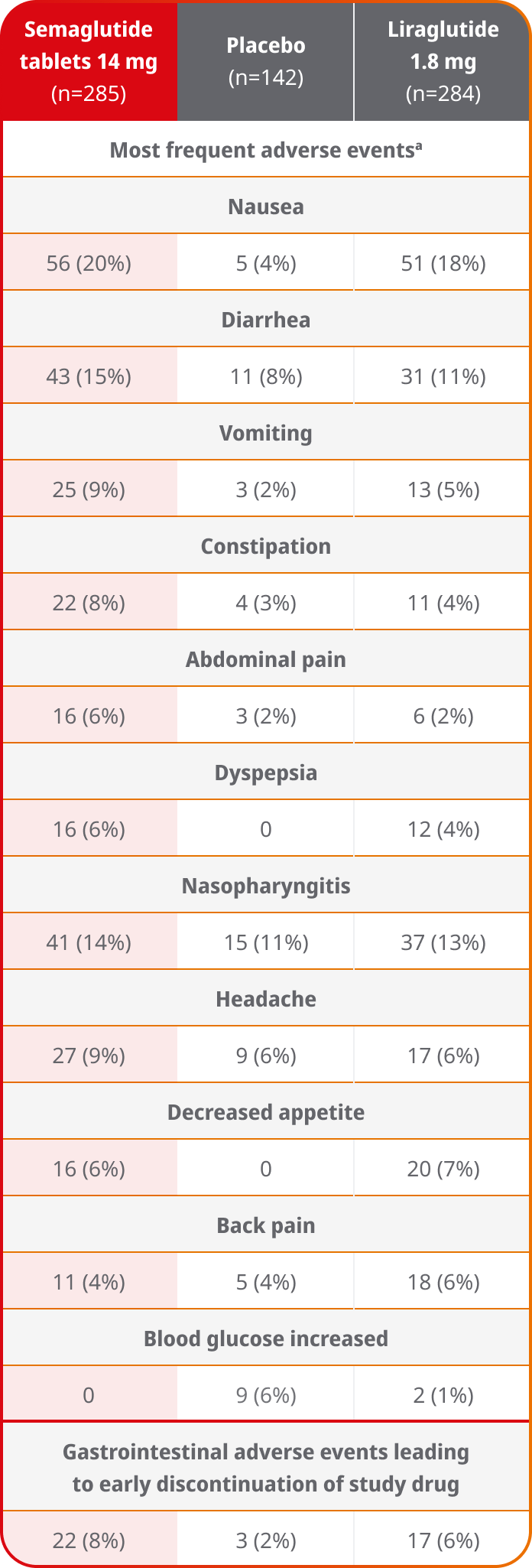
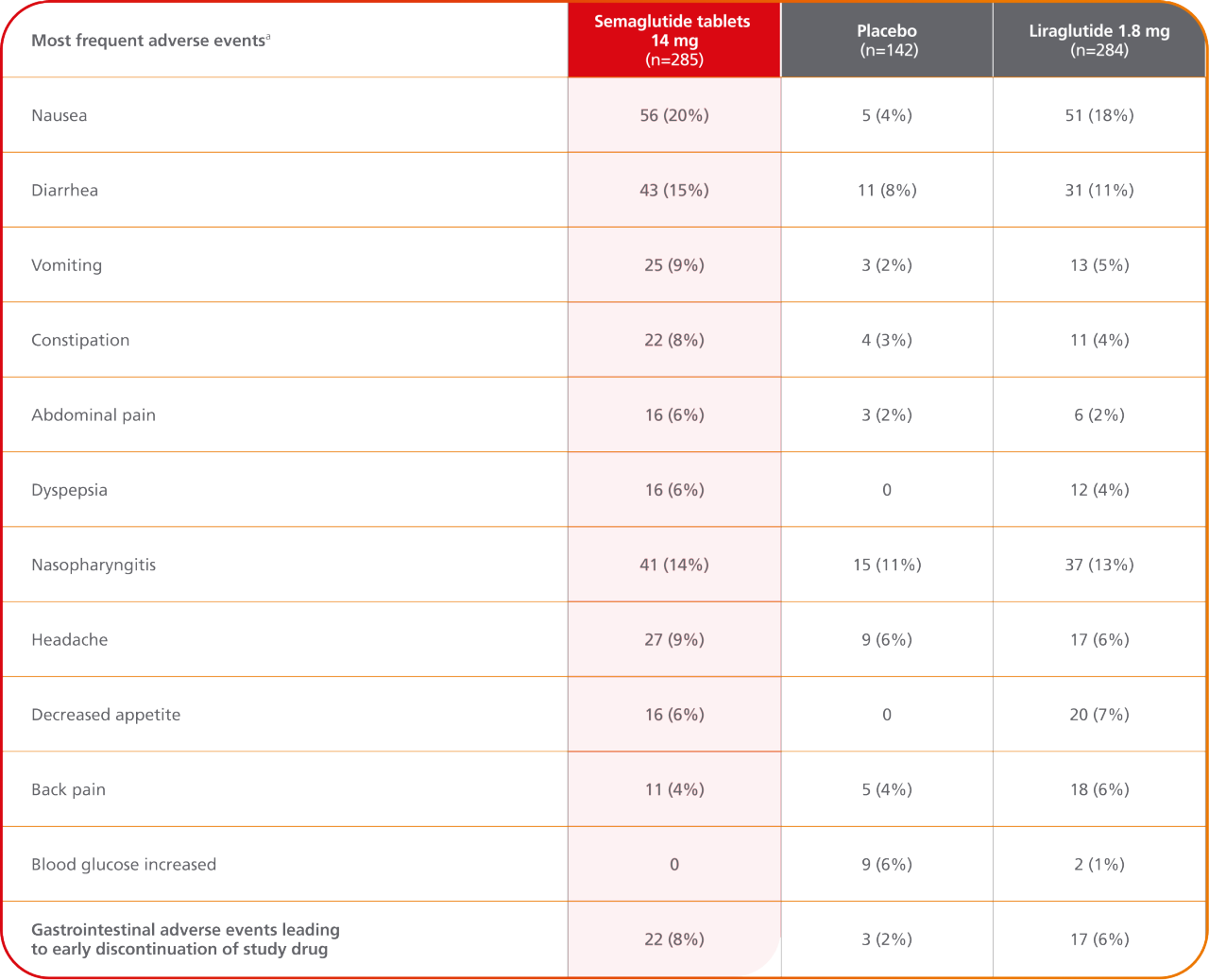
- Comparator AE rates are not an adequate basis for comparison of safety between products
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice
aData are n (%).
AE=adverse event.


STUDY DESIGNS
SUSTAIN 4: Head-to-head vs Lantus® (insulin glargine U-100)1,5
Study design: 30-week, randomized, open-label, active-controlled, parallel-group, multinational, multicenter trial to evaluate the efficacy and safety of Ozempic® injection vs insulin glargine U-100.
Patients: A total of 1089 insulin-naïve adult patients with T2D inadequately controlled on metformin alone (48%) or in combination with sulfonylurea (51%) were randomized to receive once-weekly Ozempic® injection 0.5 mg (n=362), once-weekly Ozempic® injection 1 mg (n=360), or once-daily insulin glargine U-100 (n=360). Patients assigned to insulin glargine had a baseline mean A1C of 8.1% and were started on a dose of 10 units once daily. Insulin glargine dose adjustments occurred throughout the trial period based on self-measured fasting plasma glucose before breakfast, targeting 71 to <100 mg/dL. In addition, investigators could titrate insulin glargine based on their discretion between study visits. Twenty-six percent of patients had been titrated to goal by the primary endpoint at Week 30, at which time the mean daily insulin dose was 29 units per day.
Primary endpoint: Mean change in A1C from baseline at Week 30.
Secondary endpoints: Mean change in body weight from baseline at Week 30; proportion of patients achieving A1C <7% at Week 30.
SUSTAIN 5: As an add-on to basal insulin vs placebo1,6
Study design: 30-week, randomized, double-blind, placebo-controlled, parallel-group, multinational, multicenter trial to compare the efficacy and safety of Ozempic® injection in combination with basal insulin vs volume-matched placebo in combination with basal insulin.
Patients: A total of 397 adult patients with T2D inadequately controlled on basal insulin with or without metformin were randomized to once-weekly Ozempic® injection 0.5 mg (n=132), Ozempic® injection 1 mg (n=131), or placebo (n=133). Randomization was stratified according to A1C at screening. Patients with A1C ≤8% at screening reduced the insulin dose by 20% at the start of the trial to reduce the risk of hypoglycemia.
Primary endpoint: Mean change in A1C from baseline at Week 30.
Secondary endpoints: Mean change in body weight from baseline at Week 30; proportion of patients achieving A1C <7% at Week 30; change in mean fasting plasma glucose (FPG) at Week 30.
SUSTAIN 6: Cardiovascular outcomes trial1,8
Study design: 2-year, randomized, multicenter, multinational, placebo-controlled, double-blind cardiovascular outcomes trial designed to assess noninferiority of Ozempic® injection vs placebo, both in addition to standard of care, for time to first MACE using a risk margin of 1.3.
Patients: A total of 3297 adult patients with inadequately controlled T2D and atherosclerotic cardiovascular disease were randomized to once-weekly Ozempic® injection 0.5 mg (n=826), Ozempic® injection 1 mg (n=822), or placebo (n=1649), all in addition to standard of care treatments for diabetes and CVD, such as oral antidiabetic treatments, insulin, antihypertensives, diuretics, lipid-lowering therapies, and antithrombotic medications at investigator discretion.
Primary composite endpoint: Time from randomization to first occurrence of a 3-part composite MACE, defined as CV death, nonfatal MI, or nonfatal stroke.
Secondary endpoints: Time from randomization to event onset for each of the following components of the 3-part composite MACE: CV death, nonfatal MI, and nonfatal stroke.
SUSTAIN 7: Head-to-head vs Trulicity® (dulaglutide)9
Study design: 40-week, multinational, multicenter, randomized, open-label, 4-armed, pairwise, active-controlled, parallel-group trial to compare the efficacy and safety of Ozempic® injection vs dulaglutide.
Patients: A total of 1201 adult patients with T2D inadequately controlled on metformin were randomized to receive Ozempic® injection 0.5 mg (n=301), Ozempic® injection 1 mg (n=300), dulaglutide 0.75 mg (n=299), or dulaglutide 1.5 mg (n=299) once weekly.
Primary endpoint: Mean change in A1C from baseline at Week 40.
Secondary endpoints: Mean change in body weight from baseline at Week 40; proportion of patients achieving A1C <7% at Week 40.
SUSTAIN FORTE: Ozempic® injection 1 mg vs 2 mg1,3
Study design: 40-week, randomized, active-controlled, parallel-group, double-blind, phase 3B efficacy and safety trial of Ozempic® injection 2 mg vs Ozempic® injection 1 mg in patients with T2D in need of treatment intensification.
Patients: A total of 961 adult patients with inadequately controlled T2D (A1C 8.0%-10.0%) on metformin with or without a sulfonylurea were randomized 1:1 to 2 mg (n=480) or 1 mg (n=481) of once-weekly Ozempic® injection.
Primary endpoint: Mean change in A1C from baseline at Week 40.
Secondary endpoints: Mean change in body weight from baseline at Week 40; proportion of patients achieving A1C <7% at Week 40.
FLOW: Kidney outcomes trial1,10
Study design: A randomized, double-blind, placebo-controlled, event-driven trial in adults with T2D and CKD. Median duration of follow-up was 41 months.
Patients: A total of 3533 adult patients with a mean eGFR of 47 mL/min/1.73 m2 (11% of patients having an eGFR <30 mL/min/1.73 m2, and median UACR 568 mg/g with 69% of patients with UACR >300 mg/g at baseline) were randomized 1:1 to receive Ozempic® injection 1 mg (n=1767) or placebo (n=1766) once-weekly, both in addition to standard-of-care treatments, including a maximum tolerated labeled dose of a renin-angiotensin-aldosterone system (RAAS) blocking agent, including an angiotensin converting enzyme (ACE) inhibitor or an angiotensin II receptor blocker (ARB), unless such treatment was contraindicated or not tolerated. At baseline, 95% of patients were treated with an ACE inhibitor or ARB, 16% were on SGLT-2is, 76% were on a statin, and 50% were on an antiplatelet agent.
Primary composite endpoint: Time to first occurrence of ≥50% sustained (≥28 days) eGFR decline, kidney failure (sustained eGFR <15 mL/min/1.73 m2, or initiation of long-term dialysis or kidney transplant), or renal or CV death.
Confirmatory secondary endpoints: Total eGFR slope (the annual rate of change in eGFR); MACE (a composite of nonfatal MI, nonfatal stroke, and death from CV causes), assessed in a time-to-first-event analysis; and death from any cause.
PIONEER 4: Head-to-head vs liraglutide 1.8 mg11
Study design: 52-week, randomized, double-blind, double-dummy, active controlled, and placebo-controlled phase 3a trial designed to assess the safety and efficacy of semaglutide tablets vs subcutaneously injected GLP-1 RA, liraglutide.
Patients: 711 adult patients with T2D receiving metformin alone or metformin with an SGLT-2i were randomized to semaglutide tablets 14 mg (n=285), liraglutide 1.8 mg subcutaneous injection (n=284), or placebo (n=142), all once daily.
Primary endpoint: Change in A1C from baseline to Week 26.
Confirmatory secondary endpoint: Change in body weight from baseline to Week 26.
Supportive secondary endpoints: Proportions of patients achieving A1C <7% at Week 26 and Week 52.
SOUL: CVOT13
Study design: A randomized, international, double-blind, parallel-group, placebo-controlled, event-driven, superiority phase 3b trial in adults with T2D and established atherosclerotic cardiovascular disease, CKD, or both. Median duration of follow-up was 49.6 months and 49.4 months for patients randomized to semaglutide tablets and placebo, respectively.
Patients: A total of 9650 adult patients with established CV disease without CKD (56.7%), CKD only (13%), or both CV disease and CKD (27%) were randomized 1:1 to receive semaglutide tablets 14 mg (n=4825) or placebo (n=4825) once daily, both in addition to standard-of-care treatments for glycemic control and CV risk factors including blood pressure medication, lipid-lowering agents, and antiplatelet/anticoagulant therapy. At baseline, most patients (97%) were treated with one or more glucose-lowering medications, including metformin (76%), insulin (51%), SGLT-2i (27%), sulfonylurea (29%), and DPP-4i (23%) with CV disease and risk factors managed with lipid-lowering medications (89%), platelet-aggregation inhibitors including aspirin (77%), angiotensin converting enzyme inhibitors or angiotensin II receptor blockers (79%), and beta blockers (64%). Patients received oral semaglutide or matching placebo at 3 mg for 4 weeks, followed by 7 mg for 4 weeks, then 14 mg for the remainder of the study.
Primary composite endpoint: Time to first occurrence of MACE (a composite of CV death, nonfatal MI, and nonfatal stroke).
More ways to help your patients with T2D
CKD=chronic kidney disease; CV=cardiovascular; CVD=cardiovascular disease; CVOT=cardiovascular outcomes trial; DPP-4i=dipeptidyl peptidase-4 inhibitors; eGFR=estimated glomerular filtration rate; GI=gastrointestinal; GLP-1 RA=glucagon-like peptide-1 receptor agonist; MACE=major adverse cardiovascular event; MI=myocardial infarction; SGLT-2i=sodium-glucose co-transporter-2 inhibitor; T2D=type 2 diabetes; UACR=urine albumin-to-creatinine ratio.
Important Safety Information for Ozempic®
WARNING: RISK OF THYROID C-CELL TUMORS
- In rodents, semaglutide causes dose-dependent and treatment-duration- dependent thyroid C-cell tumors at clinically relevant exposures. It is unknown whether Ozempic® causes thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans as human relevance of semaglutide-induced rodent thyroid C-cell tumors has not been determined
- Ozempic® is contraindicated in patients with a personal or family history of MTC or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Counsel patients regarding the potential risk for MTC with the use of Ozempic® and inform them of symptoms of thyroid tumors (e.g., a mass in the neck, dysphagia, dyspnea, persistent hoarseness). Routine monitoring of serum calcitonin or using thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with Ozempic®
Contraindications
- Ozempic® is contraindicated in patients with a personal or family history of MTC or in patients with MEN 2, and in patients with a prior serious hypersensitivity reaction to semaglutide or to any of the excipients in Ozempic®. Serious hypersensitivity reactions including anaphylaxis and angioedema have been reported with Ozempic®
Warnings and Precautions
- Risk of Thyroid C-Cell Tumors: Patients should be further evaluated if serum calcitonin is measured and found to be elevated or thyroid nodules are noted on physical examination or neck imaging
- Acute Pancreatitis: Acute pancreatitis, including fatal and nonfatal hemorrhagic or necrotizing pancreatitis, has been observed in patients treated with GLP-1 receptor agonists, including semaglutide. Observe patients carefully for signs and symptoms of acute pancreatitis, which may include persistent or severe abdominal pain (sometimes radiating to the back), with or without nausea or vomiting. If pancreatitis is suspected, discontinue Ozempic® and initiate appropriate management
- Diabetic Retinopathy Complications: In a 2-year trial involving patients with type 2 diabetes and high cardiovascular risk, more events of diabetic retinopathy complications occurred in patients treated with Ozempic® injection (3%) compared to placebo (1.8%). In a pooled analysis of glycemic control trials, patients reported diabetic retinopathy related adverse reactions during the trial (4.2% with semaglutide tablets and 3.8% with comparator). The absolute risk increase for diabetic retinopathy complications was larger among patients with a history of diabetic retinopathy at baseline than among patients without a known history.
Rapid improvement in glucose control has been associated with a temporary worsening of diabetic retinopathy. Patients with a history of diabetic retinopathy should be monitored for progression of diabetic retinopathy - Never Share an Ozempic® Pen Between Patients: Ozempic® pens must never be shared between patients, even if the needle is changed. Pen-sharing poses a risk for transmission of blood-borne pathogens
- Hypoglycemia: Patients receiving Ozempic® in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin may have an increased risk of hypoglycemia, including severe hypoglycemia. Inform patients using these concomitant medications of the risk of hypoglycemia and educate them on the signs and symptoms of hypoglycemia
- Acute Kidney Injury Due to Volume Depletion: There have been postmarketing reports of acute kidney injury, in some cases requiring hemodialysis, in patients treated with semaglutide. The majority of reported events occurred in patients who experienced gastrointestinal reactions leading to dehydration such as nausea, vomiting, or diarrhea. Monitor renal function in patients reporting adverse reactions to Ozempic® that could lead to volume depletion, especially during dosage initiation and escalation
- Severe Gastrointestinal (GI) Adverse Reactions: Use of Ozempic® has been associated with GI adverse reactions, sometimes severe. In clinical trials, severe GI adverse reactions were reported more frequently among patients who received Ozempic® injection (0.5 mg 0.4%, 1 mg 0.8%) than placebo (0%); and severe GI adverse reactions were reported more frequently among patients who received semaglutide tablets (7 mg 0.6%, 14 mg 2%) than placebo (0.3%). Severe GI adverse reactions have also been reported postmarketing with GLP-1 receptor agonists. Ozempic® is not recommended in patients with severe gastroparesis
- Hypersensitivity: Serious hypersensitivity reactions (e.g., anaphylaxis, angioedema) have been reported in patients treated with Ozempic®. If hypersensitivity reactions occur, discontinue use of Ozempic®; treat promptly per standard of care, and monitor until signs and symptoms resolve. Use caution in a patient with a history of angioedema or anaphylaxis with another GLP-1 receptor agonist
- Acute Gallbladder Disease: Acute events of gallbladder disease such as cholelithiasis or cholecystitis have been reported in GLP-1 receptor agonist trials and postmarketing. In placebo-controlled trials, cholelithiasis was reported in 1.5% and 0.4% of patients treated with Ozempic® injection 0.5 mg and 1 mg, respectively, and not reported in placebo-treated patients. In placebo-controlled trials to improve glycemic control, cholelithiasis was reported in 1% of patients treated with semaglutide tablets 7 mg. In a 4-year CV outcomes trial (Trial 7), cholelithiasis was reported in 1.1% of patients treated with semaglutide tablets 14 mg and in 0.9% of placebo-treated patients. In Trial 7, cholecystitis was reported in 1.1% treated with semaglutide tablets 14 mg and in 0.7% of placebo-treated patients. If cholelithiasis is suspected, gallbladder studies and appropriate clinical follow-up are indicated
- Pulmonary Aspiration During General Anesthesia or Deep Sedation: Ozempic® delays gastric emptying. There have been rare postmarketing reports of pulmonary aspiration in patients receiving GLP-1 receptor agonists undergoing elective surgeries or procedures requiring general anesthesia or deep sedation who had residual gastric contents despite reported adherence to preoperative fasting recommendations. Instruct patients to inform healthcare providers prior to any planned surgeries or procedures if they are taking Ozempic®
Adverse Reactions
- The most common adverse reactions reported in ≥5% of patients taking Ozempic® are nausea, vomiting, diarrhea, abdominal pain, and constipation. Decreased appetite was also reported in ≥5% of patients taking semaglutide tablets
Drug Interactions
- When initiating Ozempic®, consider reducing the dose of concomitantly administered insulin secretagogue (such as sulfonylureas) or insulin to reduce the risk of hypoglycemia
- Ozempic® causes a delay of gastric emptying and has the potential to impact the absorption of concomitantly administered oral medications. Monitor the effects of oral medications concomitantly administered with Ozempic®. Consider increased clinical or laboratory monitoring for medications that have a narrow therapeutic index or that require clinical monitoring
Use in Specific Populations
- Pregnancy: Available data with semaglutide use in pregnant women are not sufficient to determine a drug associated risk for major birth defects, miscarriage, or other adverse maternal or fetal outcomes. Discontinue Ozempic® in women at least 2 months before a planned pregnancy due to the long washout period for semaglutide
- Lactation: A clinical lactation study reported semaglutide concentrations below the lower limit of quantification in human breast milk. However, salcaprozate sodium (SNAC) and/or its metabolites are present in human milk. Because of the unknown potential for serious adverse reactions in the breastfed infant due to the possible accumulation of SNAC, an absorption enhancer for Ozempic® tablets, and because there are alternative formulations of semaglutide that do not contain SNAC that can be used during lactation, advise patients that breastfeeding is not recommended during treatment with Ozempic® tablets
Please click here for Ozempic® injection Prescribing Information, including Boxed Warning.
Please click here for Ozempic® tablets Prescribing Information, including Boxed Warning.
Indications and Usage
Ozempic® (semaglutide) injection 0.5 mg, 1 mg, or 2 mg and Ozempic® (semaglutide) tablets 4 mg or 9 mg are indicated:
- as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes
Ozempic® injection is indicated:
- to reduce the risk of major adverse cardiovascular (CV) events (CV death, nonfatal myocardial infarction, or nonfatal stroke) in adults with type 2 diabetes and established CV disease
- to reduce the risk of sustained eGFR decline, end-stage kidney disease, and cardiovascular death in adults with type 2 diabetes and chronic kidney disease
Ozempic® tablets are indicated:
- to reduce the risk of major adverse cardiovascular (CV) events (CV death, nonfatal myocardial infarction, or nonfatal stroke) in adults with type 2 diabetes who are at high risk for these events
Important Safety Information for Ozempic®
WARNING: RISK OF THYROID C-CELL TUMORS
- In rodents, semaglutide causes dose-dependent and treatment-duration- dependent thyroid C-cell tumors at clinically relevant exposures. It is unknown whether Ozempic® causes thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans as human relevance of semaglutide-induced rodent thyroid C-cell tumors has not been determined
- Ozempic® is contraindicated in patients with a personal or family history of MTC or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Counsel patients regarding the potential risk for MTC with the use of Ozempic® and inform them of symptoms of thyroid tumors (e.g., a mass in the neck, dysphagia, dyspnea, persistent hoarseness). Routine monitoring of serum calcitonin or using thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with Ozempic®
Important Safety Information for Ozempic®
WARNING: RISK OF THYROID C-CELL TUMORS
- In rodents, semaglutide causes dose-dependent and treatment-duration- dependent thyroid C-cell tumors at clinically relevant exposures. It is unknown whether Ozempic® causes thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans as human relevance of semaglutide-induced rodent thyroid C-cell tumors has not been determined
- Ozempic® is contraindicated in patients with a personal or family history of MTC or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Counsel patients regarding the potential risk for MTC with the use of Ozempic® and inform them of symptoms of thyroid tumors (e.g., a mass in the neck, dysphagia, dyspnea, persistent hoarseness). Routine monitoring of serum calcitonin or using thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with Ozempic®
Contraindications
- Ozempic® is contraindicated in patients with a personal or family history of MTC or in patients with MEN 2, and in patients with a prior serious hypersensitivity reaction to semaglutide or to any of the excipients in Ozempic®. Serious hypersensitivity reactions including anaphylaxis and angioedema have been reported with Ozempic®
Warnings and Precautions
- Risk of Thyroid C-Cell Tumors: Patients should be further evaluated if serum calcitonin is measured and found to be elevated or thyroid nodules are noted on physical examination or neck imaging
- Acute Pancreatitis: Acute pancreatitis, including fatal and nonfatal hemorrhagic or necrotizing pancreatitis, has been observed in patients treated with GLP-1 receptor agonists, including semaglutide. Observe patients carefully for signs and symptoms of acute pancreatitis, which may include persistent or severe abdominal pain (sometimes radiating to the back), with or without nausea or vomiting. If pancreatitis is suspected, discontinue Ozempic® and initiate appropriate management
- Diabetic Retinopathy Complications: In a 2-year trial involving patients with type 2 diabetes and high cardiovascular risk, more events of diabetic retinopathy complications occurred in patients treated with Ozempic® injection (3%) compared to placebo (1.8%). In a pooled analysis of glycemic control trials, patients reported diabetic retinopathy related adverse reactions during the trial (4.2% with semaglutide tablets and 3.8% with comparator). The absolute risk increase for diabetic retinopathy complications was larger among patients with a history of diabetic retinopathy at baseline than among patients without a known history.
Rapid improvement in glucose control has been associated with a temporary worsening of diabetic retinopathy. Patients with a history of diabetic retinopathy should be monitored for progression of diabetic retinopathy - Never Share an Ozempic® Pen Between Patients: Ozempic® pens must never be shared between patients, even if the needle is changed. Pen-sharing poses a risk for transmission of blood-borne pathogens
- Hypoglycemia: Patients receiving Ozempic® in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin may have an increased risk of hypoglycemia, including severe hypoglycemia. Inform patients using these concomitant medications of the risk of hypoglycemia and educate them on the signs and symptoms of hypoglycemia
- Acute Kidney Injury Due to Volume Depletion: There have been postmarketing reports of acute kidney injury, in some cases requiring hemodialysis, in patients treated with semaglutide. The majority of reported events occurred in patients who experienced gastrointestinal reactions leading to dehydration such as nausea, vomiting, or diarrhea. Monitor renal function in patients reporting adverse reactions to Ozempic® that could lead to volume depletion, especially during dosage initiation and escalation
- Severe Gastrointestinal (GI) Adverse Reactions: Use of Ozempic® has been associated with GI adverse reactions, sometimes severe. In clinical trials, severe GI adverse reactions were reported more frequently among patients who received Ozempic® injection (0.5 mg 0.4%, 1 mg 0.8%) than placebo (0%); and severe GI adverse reactions were reported more frequently among patients who received semaglutide tablets (7 mg 0.6%, 14 mg 2%) than placebo (0.3%). Severe GI adverse reactions have also been reported postmarketing with GLP-1 receptor agonists. Ozempic® is not recommended in patients with severe gastroparesis
- Hypersensitivity: Serious hypersensitivity reactions (e.g., anaphylaxis, angioedema) have been reported in patients treated with Ozempic®. If hypersensitivity reactions occur, discontinue use of Ozempic®; treat promptly per standard of care, and monitor until signs and symptoms resolve. Use caution in a patient with a history of angioedema or anaphylaxis with another GLP-1 receptor agonist
- Acute Gallbladder Disease: Acute events of gallbladder disease such as cholelithiasis or cholecystitis have been reported in GLP-1 receptor agonist trials and postmarketing. In placebo-controlled trials, cholelithiasis was reported in 1.5% and 0.4% of patients treated with Ozempic® injection 0.5 mg and 1 mg, respectively, and not reported in placebo-treated patients. In placebo-controlled trials to improve glycemic control, cholelithiasis was reported in 1% of patients treated with semaglutide tablets 7 mg. In a 4-year CV outcomes trial (Trial 7), cholelithiasis was reported in 1.1% of patients treated with semaglutide tablets 14 mg and in 0.9% of placebo-treated patients. In Trial 7, cholecystitis was reported in 1.1% treated with semaglutide tablets 14 mg and in 0.7% of placebo-treated patients. If cholelithiasis is suspected, gallbladder studies and appropriate clinical follow-up are indicated
- Pulmonary Aspiration During General Anesthesia or Deep Sedation: Ozempic® delays gastric emptying. There have been rare postmarketing reports of pulmonary aspiration in patients receiving GLP-1 receptor agonists undergoing elective surgeries or procedures requiring general anesthesia or deep sedation who had residual gastric contents despite reported adherence to preoperative fasting recommendations. Instruct patients to inform healthcare providers prior to any planned surgeries or procedures if they are taking Ozempic®
Adverse Reactions
- The most common adverse reactions reported in ≥5% of patients taking Ozempic® are nausea, vomiting, diarrhea, abdominal pain, and constipation. Decreased appetite was also reported in ≥5% of patients taking semaglutide tablets
Drug Interactions
- When initiating Ozempic®, consider reducing the dose of concomitantly administered insulin secretagogue (such as sulfonylureas) or insulin to reduce the risk of hypoglycemia
- Ozempic® causes a delay of gastric emptying and has the potential to impact the absorption of concomitantly administered oral medications. Monitor the effects of oral medications concomitantly administered with Ozempic®. Consider increased clinical or laboratory monitoring for medications that have a narrow therapeutic index or that require clinical monitoring
Use in Specific Populations
- Pregnancy: Available data with semaglutide use in pregnant women are not sufficient to determine a drug associated risk for major birth defects, miscarriage, or other adverse maternal or fetal outcomes. Discontinue Ozempic® in women at least 2 months before a planned pregnancy due to the long washout period for semaglutide
- Lactation: A clinical lactation study reported semaglutide concentrations below the lower limit of quantification in human breast milk. However, salcaprozate sodium (SNAC) and/or its metabolites are present in human milk. Because of the unknown potential for serious adverse reactions in the breastfed infant due to the possible accumulation of SNAC, an absorption enhancer for Ozempic® tablets, and because there are alternative formulations of semaglutide that do not contain SNAC that can be used during lactation, advise patients that breastfeeding is not recommended during treatment with Ozempic® tablets
Please click here for Ozempic® injection Prescribing Information, including Boxed Warning.
Please click here for Ozempic® tablets Prescribing Information, including Boxed Warning.
Indications and Usage
Ozempic® (semaglutide) injection 0.5 mg, 1 mg, or 2 mg and Ozempic® (semaglutide) tablets 4 mg or 9 mg are indicated:
- as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes
Ozempic® injection is indicated:
- to reduce the risk of major adverse cardiovascular (CV) events (CV death, nonfatal myocardial infarction, or nonfatal stroke) in adults with type 2 diabetes and established CV disease
- to reduce the risk of sustained eGFR decline, end-stage kidney disease, and cardiovascular death in adults with type 2 diabetes and chronic kidney disease
Ozempic® tablets are indicated:
- to reduce the risk of major adverse cardiovascular (CV) events (CV death, nonfatal myocardial infarction, or nonfatal stroke) in adults with type 2 diabetes who are at high risk for these events
References:
- Ozempic injection. Prescribing information. Novo Nordisk Inc.
- Ozempic tablets. Prescribing information. Novo Nordisk Inc.
- Frias JP, Auerbach P, Bajaj HS, et al. Efficacy and safety of once-weekly semaglutide 2.0 mg versus 1.0 mg in patients with type 2 diabetes (SUSTAIN FORTE): a double-blind, randomized, phase 3B trial. Lancet Diabetes Endocrinol. 2021;9(9):563-574. doi:10.1016/S2213-8587(21)00174-1
- Nielsen MS, Brøndsted L, Kankam M, et al. A bioequivalence study of two formulations of oral semaglutide in healthy participants. Diabetes Ther. 2025;16(2):269-287. doi:10.1007/s13300-024-01674-8
- Aroda VR, Bain SC, Cariou B, et al. Efficacy and safety of once-weekly semaglutide versus once-daily insulin glargine as add-on to metformin (with or without sulfonylureas) in insulin-naive patients with type 2 diabetes (SUSTAIN 4): a randomised, open-label, parallel-group, multicentre, multinational, phase 3a trial. Lancet Diabetes Endocrinol. 2017;5(5):355-366. doi:10.1016/S2213-8587(17)30085-2
- Rodbard HW, Rosenstock J, Canani LH, et al; PIONEER 2 Investigators. Oral semaglutide versus empagliflozin in patients with type 2 diabetes uncontrolled on metformin: the PIONEER 2 trial. Diabetes Care. 2019;42(12):2272-2281. doi:10.2337/dc19-0883
- Data on file. Novo Nordisk Inc; Plainsboro, NJ.
- Marso SP, Bain SC, Consoli A, et al; SUSTAIN-6 Investigators. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375(19):1834-1844. doi:10.1056/NEJMoa1607141
- Pratley RE, Aroda VR, Lingvay I, et al; SUSTAIN 7 Investigators. Semaglutide versus dulaglutide once weekly in patients with type 2 diabetes (SUSTAIN 7): a randomised, open-label, phase 3b trial. Lancet Diabetes Endocrinol. 2018;6(4):275-286. doi:10.1016/S2213-8587(18)30024-X
- Perkovic V, Tuttle KR, Rossing P, et al; FLOW Trial Committees and Investigators. Effects of semaglutide on chronic kidney disease in patients with type 2 diabetes. N Engl J Med. 2024;391(2):109-121. doi:10.1056/NEJMoa2403347
- Pratley R, Amod A, Hoff ST, et al; PIONEER 4 Investigators. Oral semaglutide versus subcutaneous liraglutide and placebo in type 2 diabetes (PIONEER 4): a randomized, double-blind, phase 3a trial. Lancet. 2019;394(10192):39-50. doi:10.1016/S0140-6736(19)31271-1
- McGuire DK, Marx N, Mulvagh SL, et al; SOUL Study Group. Oral semaglutide and cardiovascular outcomes in high-risk type 2 diabetes. Supplementary appendix. N Engl J Med. 2025;392(20):2001-2012. doi:10.1056/NEJMoa2501006
- McGuire DK, Marx N, Mulvagh SL, et al; SOUL Study Group. Oral semaglutide and cardiovascular outcomes in high-risk type 2 diabetes. N Engl J Med. 2025;392(20):2001-2012. doi:10.1056/NEJMoa2501006