Ozempic® (semaglutide) administration
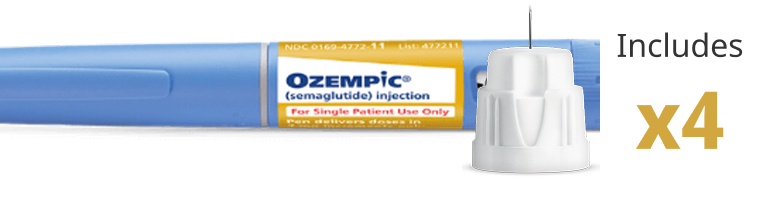
Thinnest needle (32G, 4 mm) on a once-weekly GLP-1 RA included with each pen1
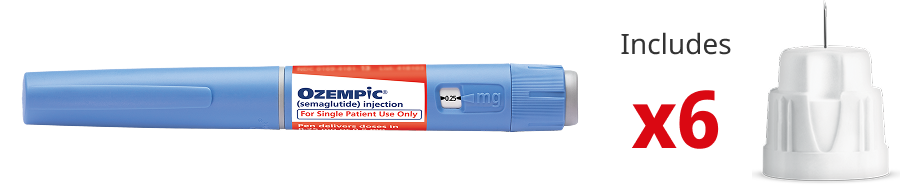
Red Label
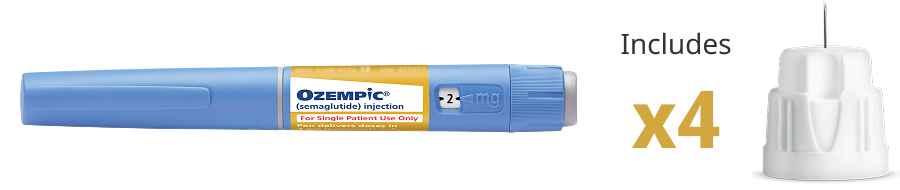
Pen that delivers doses of 0.25 mg and 0.5 mg
Contains 2 mg of Ozempic®
Delivers only the starting dose of 0.25 mg and a maintenance dose
of 0.5 mg

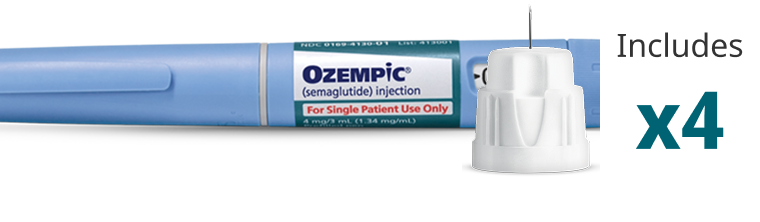
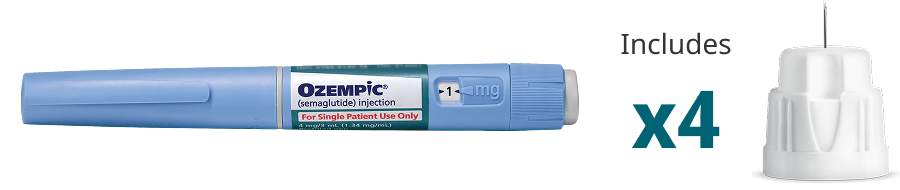
Blue Label
Pen that delivers doses of 1 mg
Contains 4 mg of Ozempic®
Delivers only the maintenance dose of 1 mg
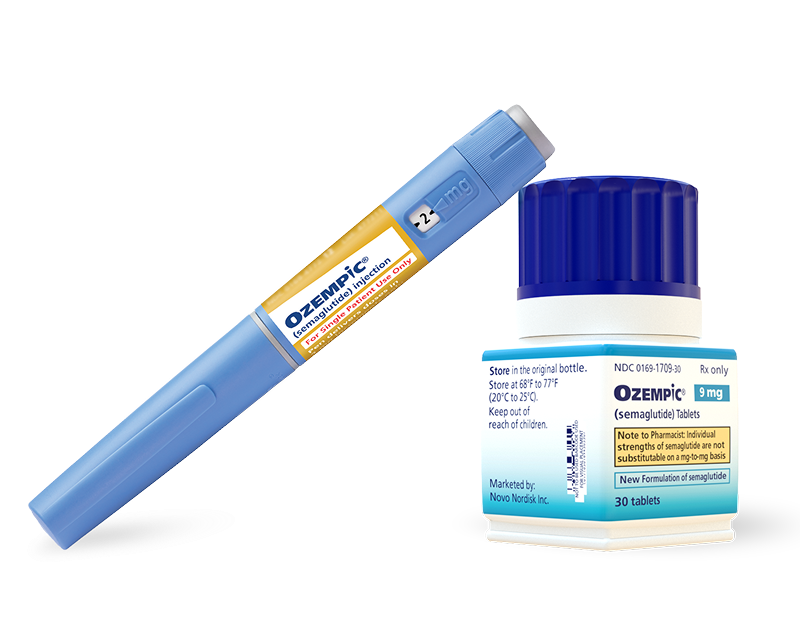
Yellow Label
Pen that delivers doses of 2 mg
Contains 8 mg of Ozempic®
Delivers only the maximum maintenance dose of 2 mg
Steps to deliver your dose1
No matter if a patient is initiating, maintaining, or escalating their Ozempic® pen dose, inform them to follow these steps for proper administration. Instruct your patients to use a new needle for each injection and to never share pens to avoid contamination or infection. The day of weekly administration can be changed if necessary as long as the time between 2 doses is at least 2 days (>48 hours).
Prepare
- Wash hands with soap and water
- Check the name and colored label on the pen and ensure the Ozempic® medicine is clear and colorless
- Take a new needle and push onto the pen, and turn to tighten
- Always use a new needle with each injection. Instruct patients to never reuse or share needles with other people. Never use a bent or damaged needle


Select
- For first-time use with each new pen, check the flow by turning the selector until the dose counter shows the flow-check symbol () then press and hold the dose button until a drop appears at the needle tip
- Only check the Ozempic® flow on the first-time use of each new pen
- Turn the dose selector until the dose counter stops and shows the correct prescribed dose


Inject
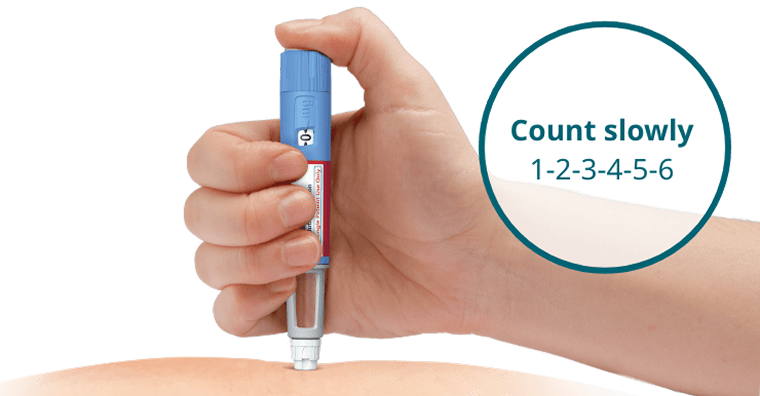
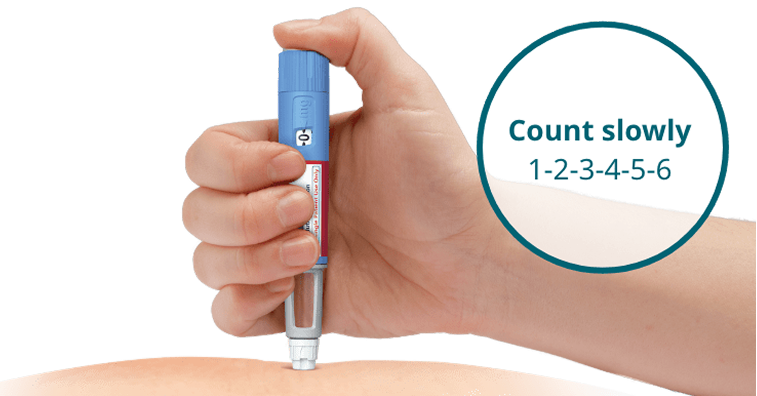
- Insert the needle into the skin and make sure you can see the dose counter
- Press and hold down the dose button until the dose counter shows 0, and continue pressing the dose button while keeping the needle in the skin
- Count 6 seconds while keeping the dose button pressed. If the needle is removed earlier, a stream of Ozempic® may come from the needle tip. If this happens, the full dose will not be delivered
- After injection, carefully remove the needle and dispose of it properly, then put the cap back on the pen and store properly
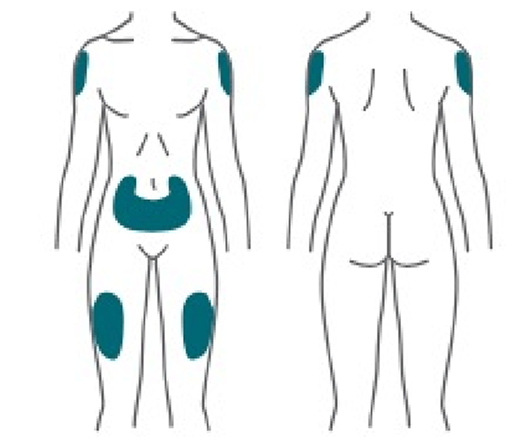
Using the pen
Ozempic® is injected under the skin of the abdomen, thigh, or upper arm (NOT into a muscle or vein). Patients should change (rotate) the injection site with each injection and not use the same site for each injection. Tell your patients to choose the injection site and wipe the skin with an alcohol swab and to let the injection site dry before they inject their dose.
How to store the Ozempic® Pen1
Prior to first use
Until expiration date
Refrigerated
36 °F to 46 °F (2 °C to 8 °C)
After first use
56 days
Room Temperature
59 °F to 86 °F (15 °C to 30 °C)
Refrigerated
36 °F to 46 °F (2 °C to 8 °C)
Learn how to prescribe Ozempic® pen
Where should the Ozempic® pen be injected?1
Ozempic® pen is injected under the skin of the abdomen, thigh, or upper arm (NOT into a muscle or vein). Patients should change (rotate) the injection site with each injection and not use the same site for each injection. Tell your patients to choose the injection site and wipe the skin with an alcohol swab and to let the injection site dry before they inject their dose.
How can patients tell their Ozempic® pen is empty?1
Patients can tell their Ozempic® pen is empty by observing the dose counter. When the Ozempic® pen is empty, the dose counter on the pen shows "0". This indicates that the pen has delivered all its pre-filled doses.
How should patients store their Ozempic® pen?1
Unused Ozempic® pens should always be stored in the refrigerator between 36° F to 46° F (2° C to 8° C). Patients should keep them in their original carton to protect them from light until the first use. Unused pens, when stored correctly in the refrigerator, can be used until the expiration date ("EXP") printed on the label.
Once patients begin using an Ozempic® pen, they have more flexibility. The opened pen can be stored either at room temperature between 59º F and 86º F (15º C to 30º C), or kept in the refrigerator (36° F to 46° F or 2° C to 8° C). Regardless of where Ozempic® pen is stored after first use, patients must dispose of the pen after 56 days, even if there is still medicine left inside. This 56-day limit ensures the medication's stability and sterility. Patients should always keep the pen cap on to protect it from light.
It is critical to never freeze Ozempic®. Patients should not use any Ozempic® pen that has been frozen, even if it has thawed, as freezing can damage the medication. Also, patients should avoid leaving pens in locations exposed to extreme temperatures, such as in a car, where they can become too hot or too cold.
How should patients dispose of their Ozempic® pen after use?1
Instruct patients that the Ozempic® pen they are using should be disposed of (thrown away) after 56 days, even if it still has Ozempic® left in it.
Instruct patients to:
Put used Ozempic® pens and needles in an FDA-cleared sharps disposal container right away after use.
If they do not have an FDA-cleared sharps disposal container, suggest that they use a household container that is:
- Made of a heavy-duty plastic
- Can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out
- Upright and stable during use
- Leak resistant
- Properly labeled to warn of hazardous waste inside the container
How should patients travel with their Ozempic® pen?1,2
When traveling with their Ozempic® pen, advise patients to:
- Transport in Carry-on: Always keep Ozempic® pen in carry-on luggage, not checked baggage, to mitigate extreme temperature fluctuations.2
- Maintain Temperature: Utilize an insulated travel case or cooler to keep the medication at a stable temperature, protecting it from direct sunlight, excessive heat, and freezing.2
- Contingency Planning: Carry spare medication to account for potential loss or damage.1
- Vehicle Travel: When traveling by car, store Ozempic® pen within the passenger cabin (not the trunk) and avoid prolonged exposure to extreme temperatures inside the vehicle.2
- Post-Travel Storage: Upon arrival, any unopened pens should be refrigerated.2
- Pre-Injection Warm-up: If refrigerated, allow the pen to reach room temperature for approximately 30 minutes before injection to enhance patient comfort.2
- Safety Check: Instruct patients not to use Ozempic® pen if it appears cloudy, contains particles, feels warm, or has been frozen.1
More ways to help your patients with T2D
GLP-1 RA=glucagon-like peptide-1 receptor agonist; T2D=type 2 diabetes.
Important Safety Information for Ozempic®
WARNING: RISK OF THYROID C-CELL TUMORS
- In rodents, semaglutide causes dose-dependent and treatment-duration- dependent thyroid C-cell tumors at clinically relevant exposures. It is unknown whether Ozempic® causes thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans as human relevance of semaglutide-induced rodent thyroid C-cell tumors has not been determined
- Ozempic® is contraindicated in patients with a personal or family history of MTC or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Counsel patients regarding the potential risk for MTC with the use of Ozempic® and inform them of symptoms of thyroid tumors (e.g., a mass in the neck, dysphagia, dyspnea, persistent hoarseness). Routine monitoring of serum calcitonin or using thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with Ozempic®
Contraindications
- Ozempic® is contraindicated in patients with a personal or family history of MTC or in patients with MEN 2, and in patients with a prior serious hypersensitivity reaction to semaglutide or to any of the excipients in Ozempic®. Serious hypersensitivity reactions including anaphylaxis and angioedema have been reported with Ozempic®
Warnings and Precautions
- Risk of Thyroid C-Cell Tumors: Patients should be further evaluated if serum calcitonin is measured and found to be elevated or thyroid nodules are noted on physical examination or neck imaging
- Acute Pancreatitis: Acute pancreatitis, including fatal and nonfatal hemorrhagic or necrotizing pancreatitis, has been observed in patients treated with GLP-1 receptor agonists, including semaglutide. Observe patients carefully for signs and symptoms of acute pancreatitis, which may include persistent or severe abdominal pain (sometimes radiating to the back), with or without nausea or vomiting. If pancreatitis is suspected, discontinue Ozempic® and initiate appropriate management
- Diabetic Retinopathy Complications: In a 2-year trial involving patients with type 2 diabetes and high cardiovascular risk, more events of diabetic retinopathy complications occurred in patients treated with Ozempic® injection (3%) compared to placebo (1.8%). In a pooled analysis of glycemic control trials, patients reported diabetic retinopathy related adverse reactions during the trial (4.2% with semaglutide tablets and 3.8% with comparator). The absolute risk increase for diabetic retinopathy complications was larger among patients with a history of diabetic retinopathy at baseline than among patients without a known history.
Rapid improvement in glucose control has been associated with a temporary worsening of diabetic retinopathy. Patients with a history of diabetic retinopathy should be monitored for progression of diabetic retinopathy - Never Share an Ozempic® Pen Between Patients: Ozempic® pens must never be shared between patients, even if the needle is changed. Pen-sharing poses a risk for transmission of blood-borne pathogens
- Hypoglycemia: Patients receiving Ozempic® in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin may have an increased risk of hypoglycemia, including severe hypoglycemia. Inform patients using these concomitant medications of the risk of hypoglycemia and educate them on the signs and symptoms of hypoglycemia
- Acute Kidney Injury Due to Volume Depletion: There have been postmarketing reports of acute kidney injury, in some cases requiring hemodialysis, in patients treated with semaglutide. The majority of reported events occurred in patients who experienced gastrointestinal reactions leading to dehydration such as nausea, vomiting, or diarrhea. Monitor renal function in patients reporting adverse reactions to Ozempic® that could lead to volume depletion, especially during dosage initiation and escalation
- Severe Gastrointestinal (GI) Adverse Reactions: Use of Ozempic® has been associated with GI adverse reactions, sometimes severe. In clinical trials, severe GI adverse reactions were reported more frequently among patients who received Ozempic® injection (0.5 mg 0.4%, 1 mg 0.8%) than placebo (0%); and severe GI adverse reactions were reported more frequently among patients who received semaglutide tablets (7 mg 0.6%, 14 mg 2%) than placebo (0.3%). Severe GI adverse reactions have also been reported postmarketing with GLP-1 receptor agonists. Ozempic® is not recommended in patients with severe gastroparesis
- Hypersensitivity: Serious hypersensitivity reactions (e.g., anaphylaxis, angioedema) have been reported in patients treated with Ozempic®. If hypersensitivity reactions occur, discontinue use of Ozempic®; treat promptly per standard of care, and monitor until signs and symptoms resolve. Use caution in a patient with a history of angioedema or anaphylaxis with another GLP-1 receptor agonist
- Acute Gallbladder Disease: Acute events of gallbladder disease such as cholelithiasis or cholecystitis have been reported in GLP-1 receptor agonist trials and postmarketing. In placebo-controlled trials, cholelithiasis was reported in 1.5% and 0.4% of patients treated with Ozempic® injection 0.5 mg and 1 mg, respectively, and not reported in placebo-treated patients. In placebo-controlled trials to improve glycemic control, cholelithiasis was reported in 1% of patients treated with semaglutide tablets 7 mg. In a 4-year CV outcomes trial (Trial 7), cholelithiasis was reported in 1.1% of patients treated with semaglutide tablets 14 mg and in 0.9% of placebo-treated patients. In Trial 7, cholecystitis was reported in 1.1% treated with semaglutide tablets 14 mg and in 0.7% of placebo-treated patients. If cholelithiasis is suspected, gallbladder studies and appropriate clinical follow-up are indicated
- Pulmonary Aspiration During General Anesthesia or Deep Sedation: Ozempic® delays gastric emptying. There have been rare postmarketing reports of pulmonary aspiration in patients receiving GLP-1 receptor agonists undergoing elective surgeries or procedures requiring general anesthesia or deep sedation who had residual gastric contents despite reported adherence to preoperative fasting recommendations. Instruct patients to inform healthcare providers prior to any planned surgeries or procedures if they are taking Ozempic®
Adverse Reactions
- The most common adverse reactions reported in ≥5% of patients taking Ozempic® are nausea, vomiting, diarrhea, abdominal pain, and constipation. Decreased appetite was also reported in ≥5% of patients taking semaglutide tablets
Drug Interactions
- When initiating Ozempic®, consider reducing the dose of concomitantly administered insulin secretagogue (such as sulfonylureas) or insulin to reduce the risk of hypoglycemia
- Ozempic® causes a delay of gastric emptying and has the potential to impact the absorption of concomitantly administered oral medications. Monitor the effects of oral medications concomitantly administered with Ozempic®. Consider increased clinical or laboratory monitoring for medications that have a narrow therapeutic index or that require clinical monitoring
Use in Specific Populations
- Pregnancy: Available data with semaglutide use in pregnant women are not sufficient to determine a drug associated risk for major birth defects, miscarriage, or other adverse maternal or fetal outcomes. Discontinue Ozempic® in women at least 2 months before a planned pregnancy due to the long washout period for semaglutide
- Lactation: A clinical lactation study reported semaglutide concentrations below the lower limit of quantification in human breast milk. However, salcaprozate sodium (SNAC) and/or its metabolites are present in human milk. Because of the unknown potential for serious adverse reactions in the breastfed infant due to the possible accumulation of SNAC, an absorption enhancer for Ozempic® tablets, and because there are alternative formulations of semaglutide that do not contain SNAC that can be used during lactation, advise patients that breastfeeding is not recommended during treatment with Ozempic® tablets
Please click here for Ozempic® injection Prescribing Information, including Boxed Warning.
Please click here for Ozempic® tablets Prescribing Information, including Boxed Warning.
Indications and Usage
Ozempic® (semaglutide) injection 0.5 mg, 1 mg, or 2 mg and Ozempic® (semaglutide) tablets 4 mg or 9 mg are indicated:
- as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes
Ozempic® injection is indicated:
- to reduce the risk of major adverse cardiovascular (CV) events (CV death, nonfatal myocardial infarction, or nonfatal stroke) in adults with type 2 diabetes and established CV disease
- to reduce the risk of sustained eGFR decline, end-stage kidney disease, and cardiovascular death in adults with type 2 diabetes and chronic kidney disease
Ozempic® tablets are indicated:
- to reduce the risk of major adverse cardiovascular (CV) events (CV death, nonfatal myocardial infarction, or nonfatal stroke) in adults with type 2 diabetes who are at high risk for these events
Important Safety Information for Ozempic®
WARNING: RISK OF THYROID C-CELL TUMORS
- In rodents, semaglutide causes dose-dependent and treatment-duration- dependent thyroid C-cell tumors at clinically relevant exposures. It is unknown whether Ozempic® causes thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans as human relevance of semaglutide-induced rodent thyroid C-cell tumors has not been determined
- Ozempic® is contraindicated in patients with a personal or family history of MTC or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Counsel patients regarding the potential risk for MTC with the use of Ozempic® and inform them of symptoms of thyroid tumors (e.g., a mass in the neck, dysphagia, dyspnea, persistent hoarseness). Routine monitoring of serum calcitonin or using thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with Ozempic®
Important Safety Information for Ozempic®
WARNING: RISK OF THYROID C-CELL TUMORS
- In rodents, semaglutide causes dose-dependent and treatment-duration- dependent thyroid C-cell tumors at clinically relevant exposures. It is unknown whether Ozempic® causes thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans as human relevance of semaglutide-induced rodent thyroid C-cell tumors has not been determined
- Ozempic® is contraindicated in patients with a personal or family history of MTC or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Counsel patients regarding the potential risk for MTC with the use of Ozempic® and inform them of symptoms of thyroid tumors (e.g., a mass in the neck, dysphagia, dyspnea, persistent hoarseness). Routine monitoring of serum calcitonin or using thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with Ozempic®
Contraindications
- Ozempic® is contraindicated in patients with a personal or family history of MTC or in patients with MEN 2, and in patients with a prior serious hypersensitivity reaction to semaglutide or to any of the excipients in Ozempic®. Serious hypersensitivity reactions including anaphylaxis and angioedema have been reported with Ozempic®
Warnings and Precautions
- Risk of Thyroid C-Cell Tumors: Patients should be further evaluated if serum calcitonin is measured and found to be elevated or thyroid nodules are noted on physical examination or neck imaging
- Acute Pancreatitis: Acute pancreatitis, including fatal and nonfatal hemorrhagic or necrotizing pancreatitis, has been observed in patients treated with GLP-1 receptor agonists, including semaglutide. Observe patients carefully for signs and symptoms of acute pancreatitis, which may include persistent or severe abdominal pain (sometimes radiating to the back), with or without nausea or vomiting. If pancreatitis is suspected, discontinue Ozempic® and initiate appropriate management
- Diabetic Retinopathy Complications: In a 2-year trial involving patients with type 2 diabetes and high cardiovascular risk, more events of diabetic retinopathy complications occurred in patients treated with Ozempic® injection (3%) compared to placebo (1.8%). In a pooled analysis of glycemic control trials, patients reported diabetic retinopathy related adverse reactions during the trial (4.2% with semaglutide tablets and 3.8% with comparator). The absolute risk increase for diabetic retinopathy complications was larger among patients with a history of diabetic retinopathy at baseline than among patients without a known history.
Rapid improvement in glucose control has been associated with a temporary worsening of diabetic retinopathy. Patients with a history of diabetic retinopathy should be monitored for progression of diabetic retinopathy - Never Share an Ozempic® Pen Between Patients: Ozempic® pens must never be shared between patients, even if the needle is changed. Pen-sharing poses a risk for transmission of blood-borne pathogens
- Hypoglycemia: Patients receiving Ozempic® in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin may have an increased risk of hypoglycemia, including severe hypoglycemia. Inform patients using these concomitant medications of the risk of hypoglycemia and educate them on the signs and symptoms of hypoglycemia
- Acute Kidney Injury Due to Volume Depletion: There have been postmarketing reports of acute kidney injury, in some cases requiring hemodialysis, in patients treated with semaglutide. The majority of reported events occurred in patients who experienced gastrointestinal reactions leading to dehydration such as nausea, vomiting, or diarrhea. Monitor renal function in patients reporting adverse reactions to Ozempic® that could lead to volume depletion, especially during dosage initiation and escalation
- Severe Gastrointestinal (GI) Adverse Reactions: Use of Ozempic® has been associated with GI adverse reactions, sometimes severe. In clinical trials, severe GI adverse reactions were reported more frequently among patients who received Ozempic® injection (0.5 mg 0.4%, 1 mg 0.8%) than placebo (0%); and severe GI adverse reactions were reported more frequently among patients who received semaglutide tablets (7 mg 0.6%, 14 mg 2%) than placebo (0.3%). Severe GI adverse reactions have also been reported postmarketing with GLP-1 receptor agonists. Ozempic® is not recommended in patients with severe gastroparesis
- Hypersensitivity: Serious hypersensitivity reactions (e.g., anaphylaxis, angioedema) have been reported in patients treated with Ozempic®. If hypersensitivity reactions occur, discontinue use of Ozempic®; treat promptly per standard of care, and monitor until signs and symptoms resolve. Use caution in a patient with a history of angioedema or anaphylaxis with another GLP-1 receptor agonist
- Acute Gallbladder Disease: Acute events of gallbladder disease such as cholelithiasis or cholecystitis have been reported in GLP-1 receptor agonist trials and postmarketing. In placebo-controlled trials, cholelithiasis was reported in 1.5% and 0.4% of patients treated with Ozempic® injection 0.5 mg and 1 mg, respectively, and not reported in placebo-treated patients. In placebo-controlled trials to improve glycemic control, cholelithiasis was reported in 1% of patients treated with semaglutide tablets 7 mg. In a 4-year CV outcomes trial (Trial 7), cholelithiasis was reported in 1.1% of patients treated with semaglutide tablets 14 mg and in 0.9% of placebo-treated patients. In Trial 7, cholecystitis was reported in 1.1% treated with semaglutide tablets 14 mg and in 0.7% of placebo-treated patients. If cholelithiasis is suspected, gallbladder studies and appropriate clinical follow-up are indicated
- Pulmonary Aspiration During General Anesthesia or Deep Sedation: Ozempic® delays gastric emptying. There have been rare postmarketing reports of pulmonary aspiration in patients receiving GLP-1 receptor agonists undergoing elective surgeries or procedures requiring general anesthesia or deep sedation who had residual gastric contents despite reported adherence to preoperative fasting recommendations. Instruct patients to inform healthcare providers prior to any planned surgeries or procedures if they are taking Ozempic®
Adverse Reactions
- The most common adverse reactions reported in ≥5% of patients taking Ozempic® are nausea, vomiting, diarrhea, abdominal pain, and constipation. Decreased appetite was also reported in ≥5% of patients taking semaglutide tablets
Drug Interactions
- When initiating Ozempic®, consider reducing the dose of concomitantly administered insulin secretagogue (such as sulfonylureas) or insulin to reduce the risk of hypoglycemia
- Ozempic® causes a delay of gastric emptying and has the potential to impact the absorption of concomitantly administered oral medications. Monitor the effects of oral medications concomitantly administered with Ozempic®. Consider increased clinical or laboratory monitoring for medications that have a narrow therapeutic index or that require clinical monitoring
Use in Specific Populations
- Pregnancy: Available data with semaglutide use in pregnant women are not sufficient to determine a drug associated risk for major birth defects, miscarriage, or other adverse maternal or fetal outcomes. Discontinue Ozempic® in women at least 2 months before a planned pregnancy due to the long washout period for semaglutide
- Lactation: A clinical lactation study reported semaglutide concentrations below the lower limit of quantification in human breast milk. However, salcaprozate sodium (SNAC) and/or its metabolites are present in human milk. Because of the unknown potential for serious adverse reactions in the breastfed infant due to the possible accumulation of SNAC, an absorption enhancer for Ozempic® tablets, and because there are alternative formulations of semaglutide that do not contain SNAC that can be used during lactation, advise patients that breastfeeding is not recommended during treatment with Ozempic® tablets
Please click here for Ozempic® injection Prescribing Information, including Boxed Warning.
Please click here for Ozempic® tablets Prescribing Information, including Boxed Warning.
Indications and Usage
Ozempic® (semaglutide) injection 0.5 mg, 1 mg, or 2 mg and Ozempic® (semaglutide) tablets 4 mg or 9 mg are indicated:
- as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes
Ozempic® injection is indicated:
- to reduce the risk of major adverse cardiovascular (CV) events (CV death, nonfatal myocardial infarction, or nonfatal stroke) in adults with type 2 diabetes and established CV disease
- to reduce the risk of sustained eGFR decline, end-stage kidney disease, and cardiovascular death in adults with type 2 diabetes and chronic kidney disease
Ozempic® tablets are indicated:
- to reduce the risk of major adverse cardiovascular (CV) events (CV death, nonfatal myocardial infarction, or nonfatal stroke) in adults with type 2 diabetes who are at high risk for these events
References:
- Ozempic injection. Prescribing information. Novo Nordisk Inc.
- How to travel with Ozempic. Healthline. Updated September 2, 2025. Accessed March 18, 2026. https://www.healthline.com/health/drugs/how-to-travel-with-ozempic
- Ozempic tablets. Prescribing information. Novo Nordisk Inc.

