For adults with T2D and established CVD
Proven CV risk reduction with Ozempic® (semaglutide) pen1


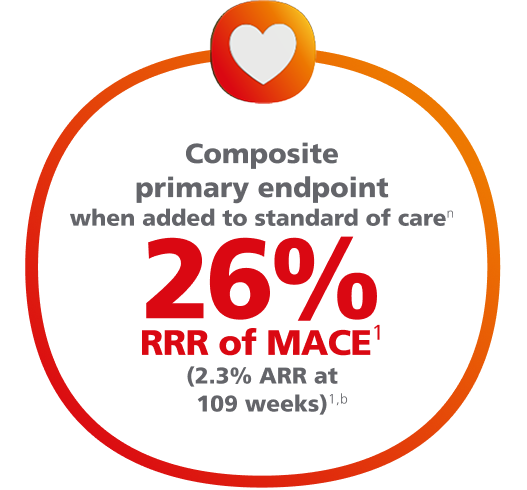
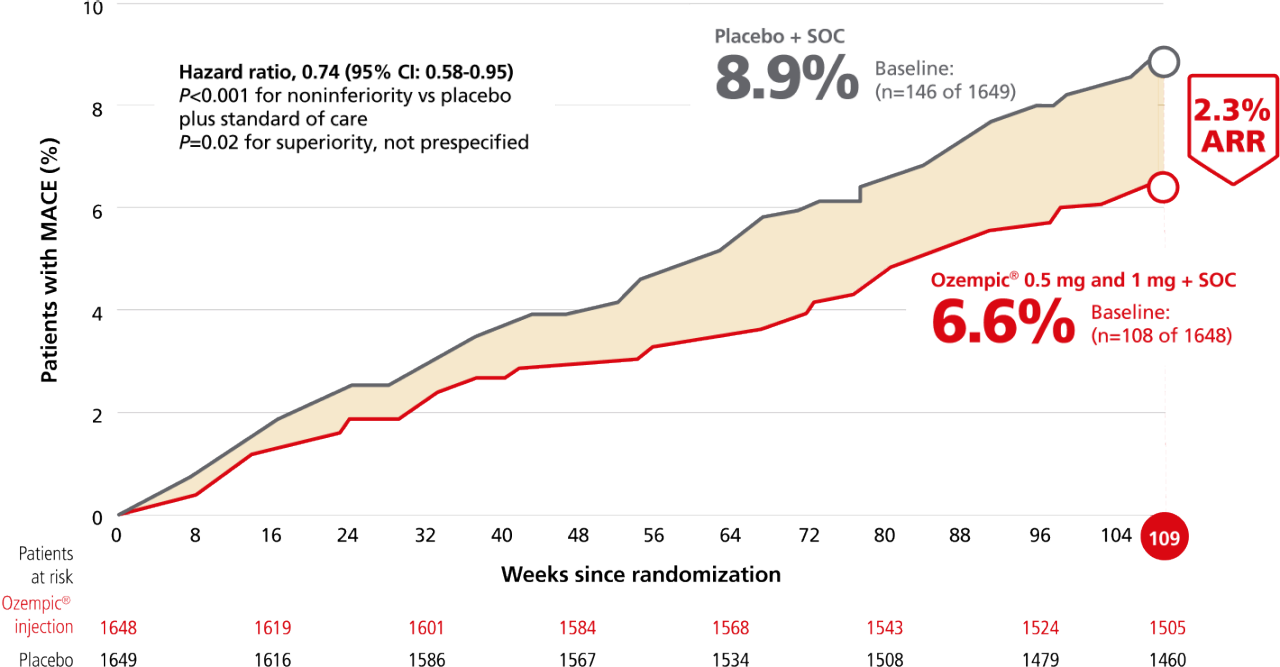
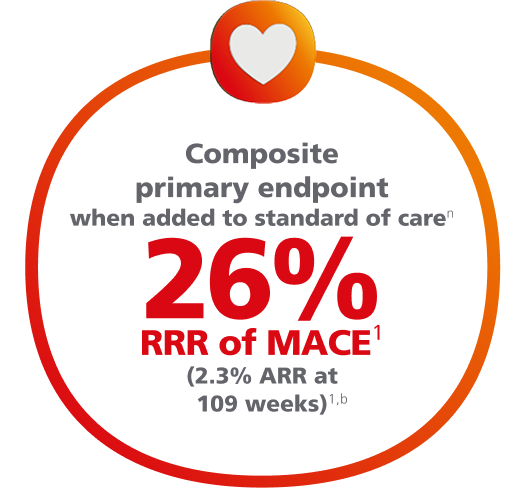
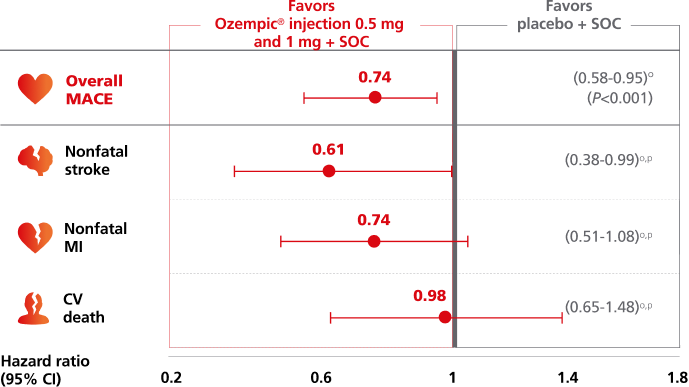
Ozempic® pen significantly lowered the risk of MACE1,2
For adults with T2D and established CVD, for the composite primary endpoint after 2 years vs placebo, when both were added to SOC1,2,a
Composite MACE endpoint: CV death, nonfatal MI, or nonfatal stroke.1
Time to first confirmed major adverse CV event (MACE)1,2,b
NNT 45
Number needed to treat to prevent
1 MACE
(2 years)c
aStandards of care included, but were not limited to, oral antidiabetic treatments, insulin, antihypertensives, diuretics, lipid-lowering therapies, and antithrombotic medications.3
bResults apply to Ozempic® 0.5 mg and 1 mg plus SOC vs placebo plus SOC.1
cEstimated cumulative risk of MACE at Week 104 was 6.2% with Ozempic® pen 0.5 mg and 1 mg and 8.4% with placebo.3
SUSTAIN 6: A 2-year CVOT for Ozempic® pen1,2
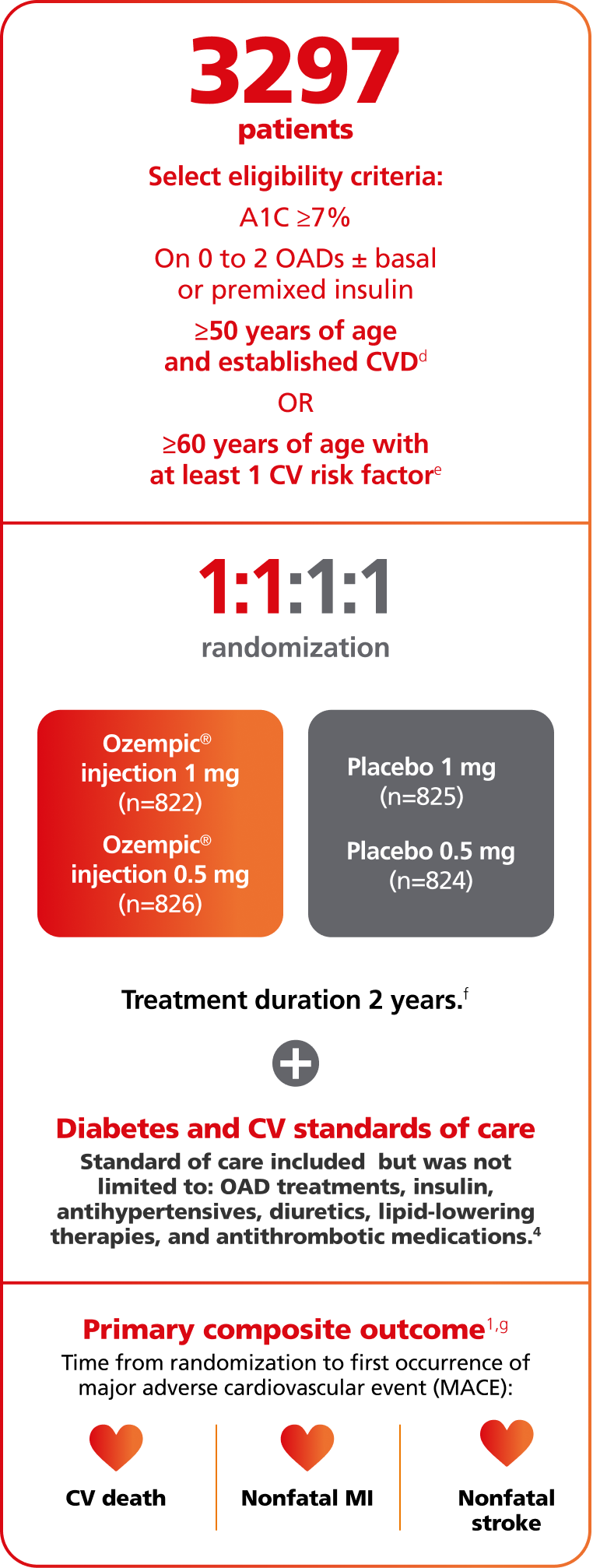
A 109-week, multicenter, multinational, placebo-controlled, double-blind CVOT in 3297 adult patients with inadequately controlled T2D and atherosclerotic cardiovascular disease. Patients had a mean duration of diabetes of 13.9 years and a mean baseline A1C of 8.7%, and 58% were taking insulin.
Additional SUSTAIN 6 study information
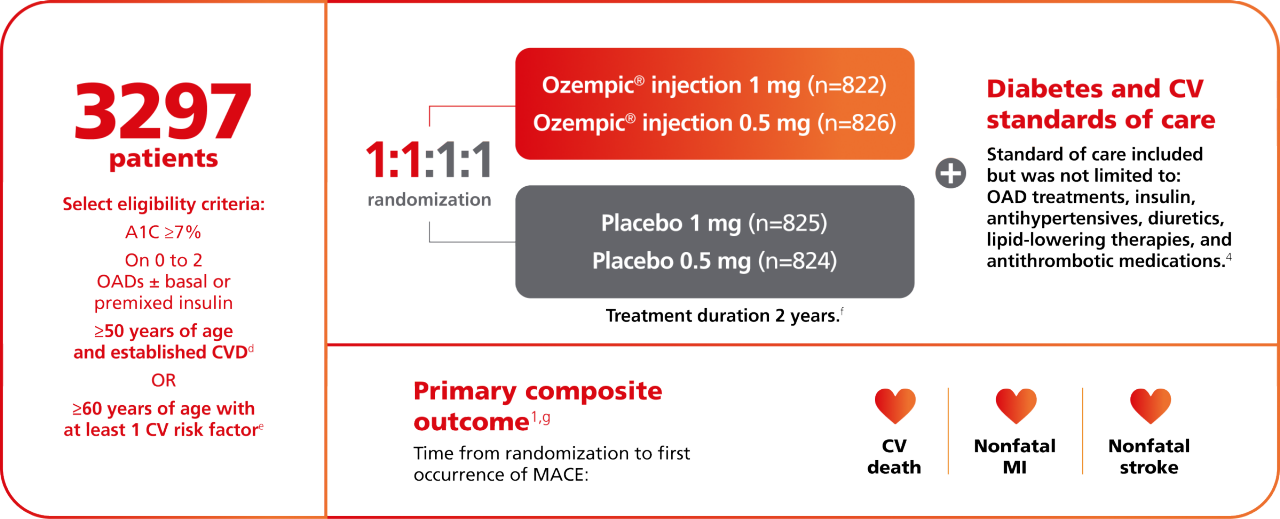
dEstablished CV disease (previous CV, cerebrovascular, or peripheral vascular disease) or chronic heart failure (New York Heart Association class II or III) or chronic kidney disease stage 3 or higher.1,2
eDefined as persistent microalbuminuria (30-299 mg/g) or proteinuria, hypertension and left ventricular hypertrophy by electrocardiogram or imaging, left ventricular systolic or diastolic dysfunction by imaging, or ankle/brachial index <0.9 of those in the trial.4
fTrial consisted of 104 weeks of treatment (including a 4- to 8-week dose escalation period), with a subsequent 5-week follow-up period.1,2
gFor the primary analysis, a Cox proportional-hazards model was used to test for noninferiority of Ozempic® pen to placebo for time to first MACE using a risk margin of 1.3.1,2
Scroll below for MACE events by weight-loss category (post hoc analysis)
83% of patients had established CVD2,h,i
Patient characteristics (n=3297)1,2


hPrior CV disease was defined as age 60 years with at least 1 CV risk factor (persistent microalbuminuria [30-299 mg/g] or proteinuria, hypertension and left ventricular hypertrophy by electrocardiogram or imaging, left ventricular systolic or diastolic dysfunction by imaging, or ankle/brachial index <0.9) or age ≥50 years with established CVD (previous CV, cerebrovascular, or peripheral vascular disease; chronic heart failure [New York Heart Association class II or III]; or CKD of stage 3 or higher).2,3
iIn total, 1940 patients (58.8%) had established CV disease without CKD, 353 (10.7%) had CKD only, and 442 (13.4%) had both CV disease and CKD; 562 patients (17%) had CV risk factors without established CV disease or CKD. In the trial, 453 patients (13.7%) had peripheral artery disease.4,5
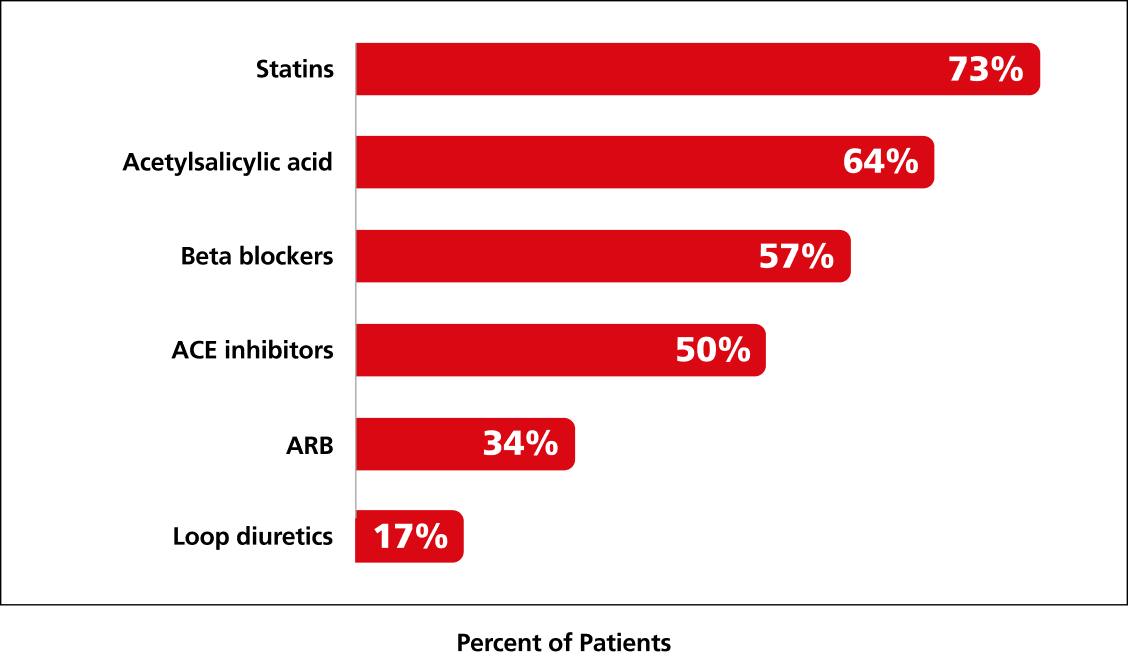
98% of patients were receiving CV medications at baseline2,4
Summary of CV medication usage at baseline (n=3297)4
History of CVD at screening (n=3297)1,4,j
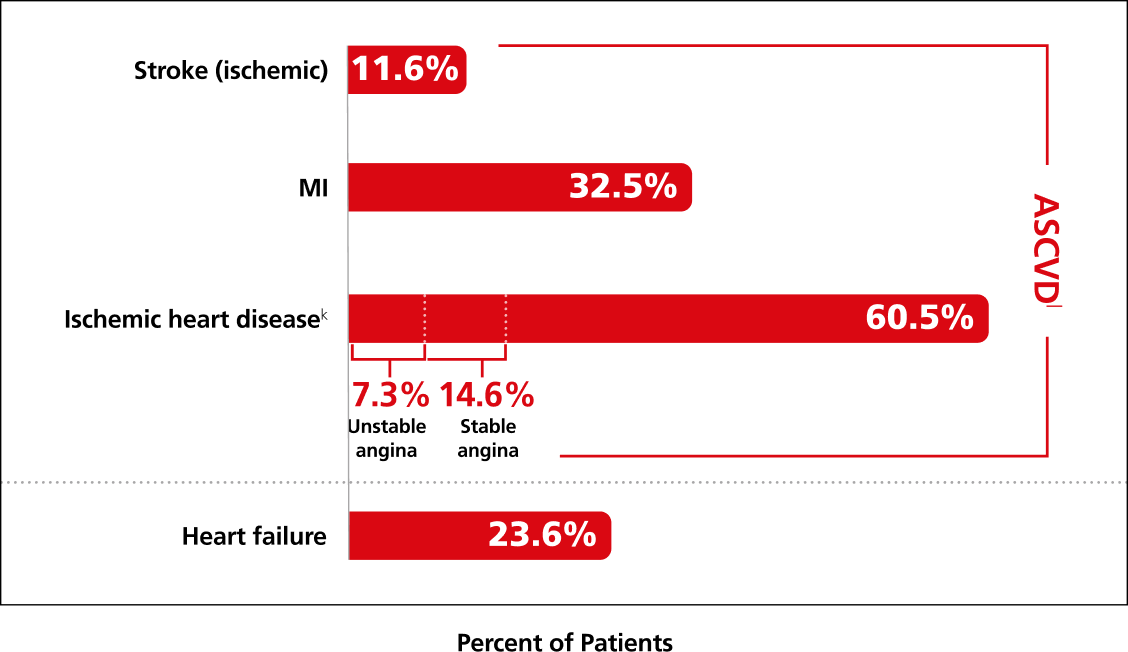
jHistory at baseline among subjects enrolled in the trial (Ozempic® pen and placebo groups).5
kComprising asymptomatic (silent) cardiac ischemia (8.6%), stable angina pectoris (14.6%), unstable angina (7.3%), non-ST-segment elevation (11%), ST-segment elevation MI (2.1%) and unknown (6.7%).3,4
lThe ACC defines ASCVD as: a history of an acute coronary syndrome or myocardial infarction, stable or unstable angina, coronary heart disease with or without revascularization, other arterial revascularization, stroke, or peripheral artery disease assumed to be atherosclerotic in origin.5
ACC=American College of Cardiology; ACE=angiotensin-converting enzyme; ARB=angiotensin receptor blocker; ASCVD=atherosclerotic cardiovascular disease; BMI=body mass index; CKD=chronic kidney disease; CV=cardiovascular; CVD=cardiovascular disease; MI=myocardial infarction.
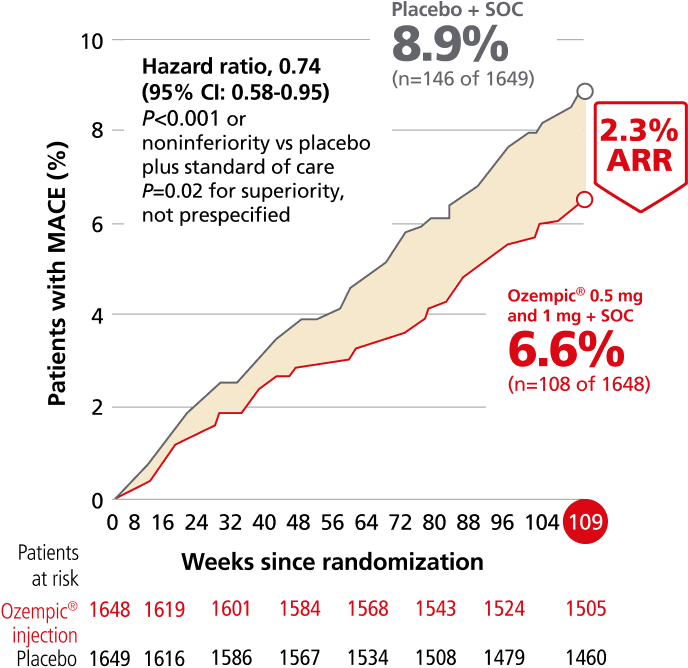
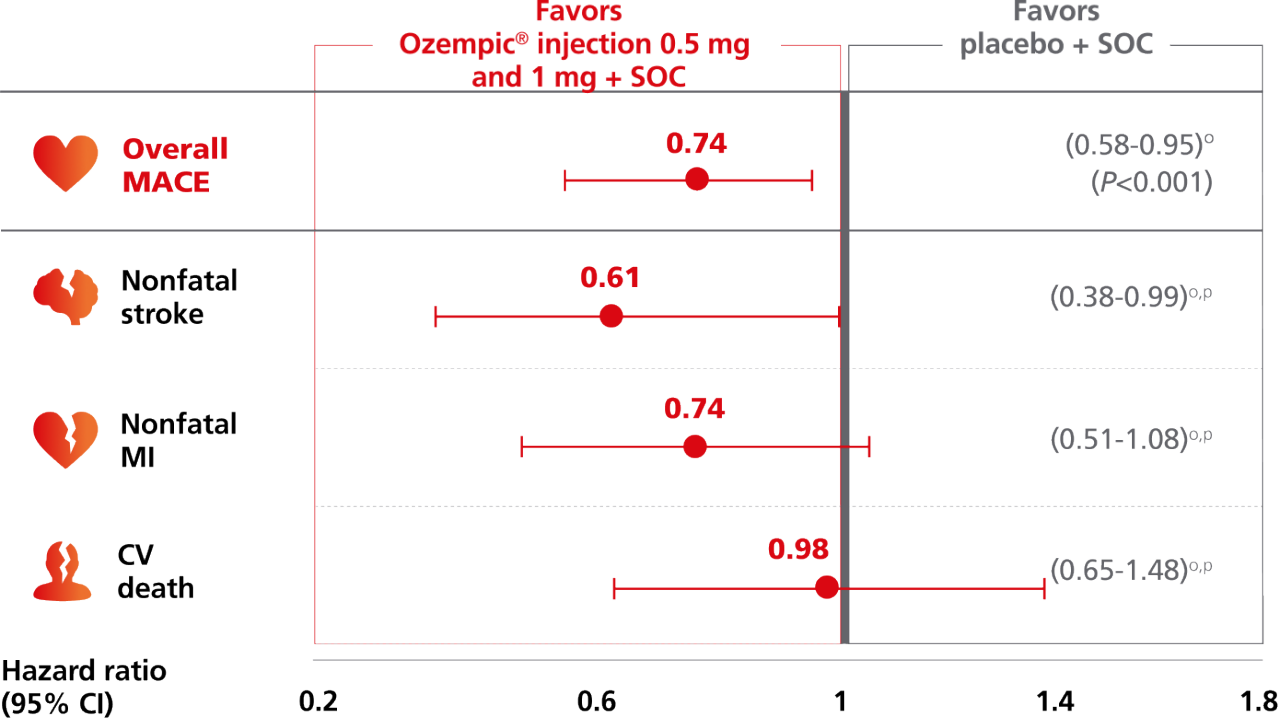
Ozempic® pen significantly reduced risk of MACE, driven by rate of nonfatal stroke1,2
For adults with T2D and established CVD, for the composite primary endpoint after 2 years vs placebo, when both were added to SOC1,m
Composite MACE endpoint: CV death, nonfatal MI,
or nonfatal stroke1,2
39%
significant
reduction
in rate of
nonfatal stroke
drove the MACE
reduction1,2,q
This study was not powered to detect a difference in the individual endpoints of the composite MACE. Absolute rates for the combined endpoint of “fatal or nonfatal stroke”: 1.8% (n=30 of 1648) with Ozempic® injection and 2.8% (n=46 of 1649) with placebo. HR, 0.65 (95% CI, 0.41-1.03); P=NS. There was no significant change in the rate of nonfatal MI or CV death.1,2
Overall MACE occurred in 108 patients of 1648 (6.6%) on Ozempic® injection (0.5 mg and 1 mg) and 146 patients of 1649 (8.9%) on placebo (HR, 0.74 [95% CI: 0.58-0.95] P<0.001 for noninferiority, P=0.02 for superiority, not prespecified); nonfatal stroke occurred in 27 patients (1.6%) on Ozempic® injection and 44 patients (2.7%) on placebo; nonfatal Ml occurred in 47 patients (2.9%) on Ozempic® injection and 64 patients (3.9%) on placebo; CV death occurred in 44 patients (2.7%) on Ozempic® injection and 46 patients (2.8%) on placebo.1,2
mStandards of care included, but were not limited to, oral antidiabetic treatments, insulin, antihypertensives, diuretics, lipid-lowering therapies, and antithrombotic medications.4
nResults apply to Ozempic® injection 0.5 mg and 1 mg plus standard of care vs placebo plus standard of care.1
oHazard ratio vs placebo (95% CI). Median study observation time of 2.1 years. Cox proportional-hazards models with treatment as fixed factor and stratified by evidence of cardiovascular disease, insulin treatment, and renal impairment. P values other than for the primary hypothesis (noninferiority) are unadjusted for multiplicity and test null hypotheses of no difference.1,2
pP value is not adjusted for multiplicity and tests null hypotheses of no difference (post hoc).2
qThe primary endpoint in the SUSTAIN 6 CVOT was time to first occurrence of a 3-part composite MACE that included CV death, nonfatal MI, or nonfatal stroke.1
This analysis is descriptive and observational only and not intended to inform clinical decisions.
The clinical validity of these results cannot be determined.
This post hoc exploratory subgroup analysis was not prespecified in the trial, and the trial was neither designed nor powered for this analysis. As such, the analysis is hypothesis-generating, not hypothesis-testing.
These data were analyzed by Novo Nordisk and the results are not published.


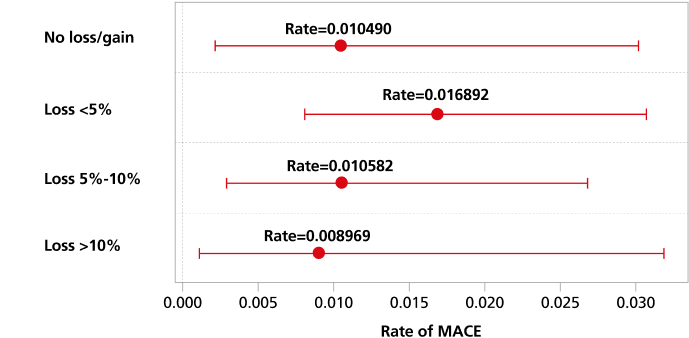
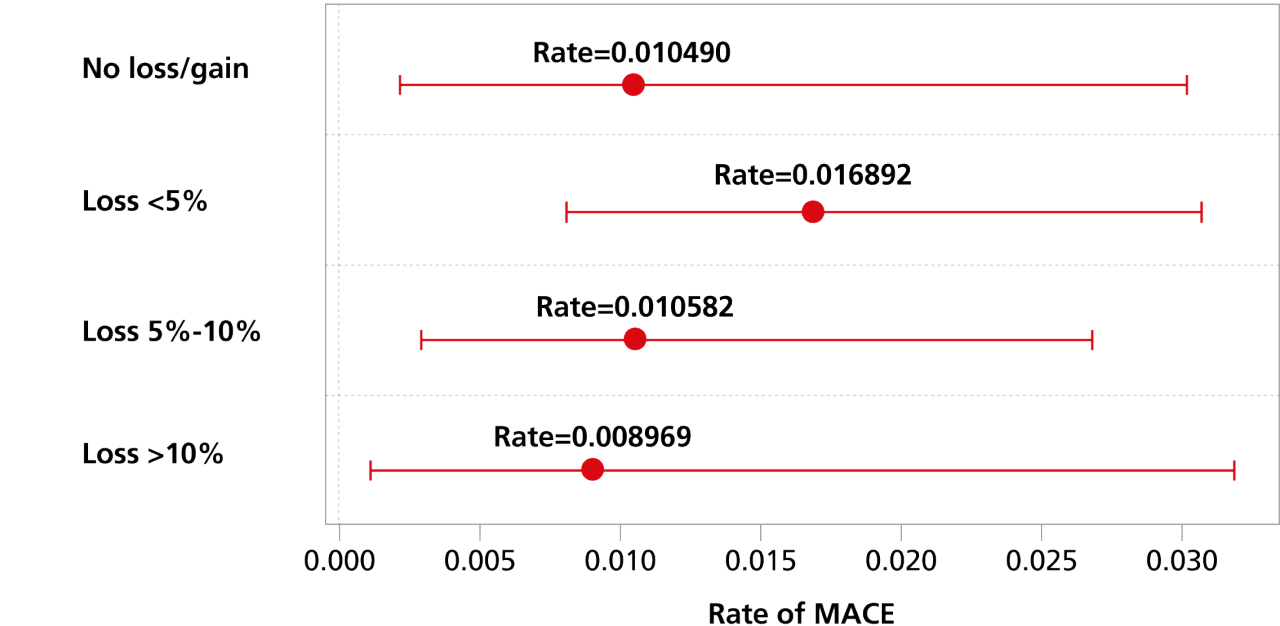
MACE by weight-loss category were assessed3
Change in body weight from baseline to Week 104 was a secondary endpoint in SUSTAIN 6.
Ozempic® is not indicated for weight loss
Rate of MACE events by weight-loss category with 95% CI
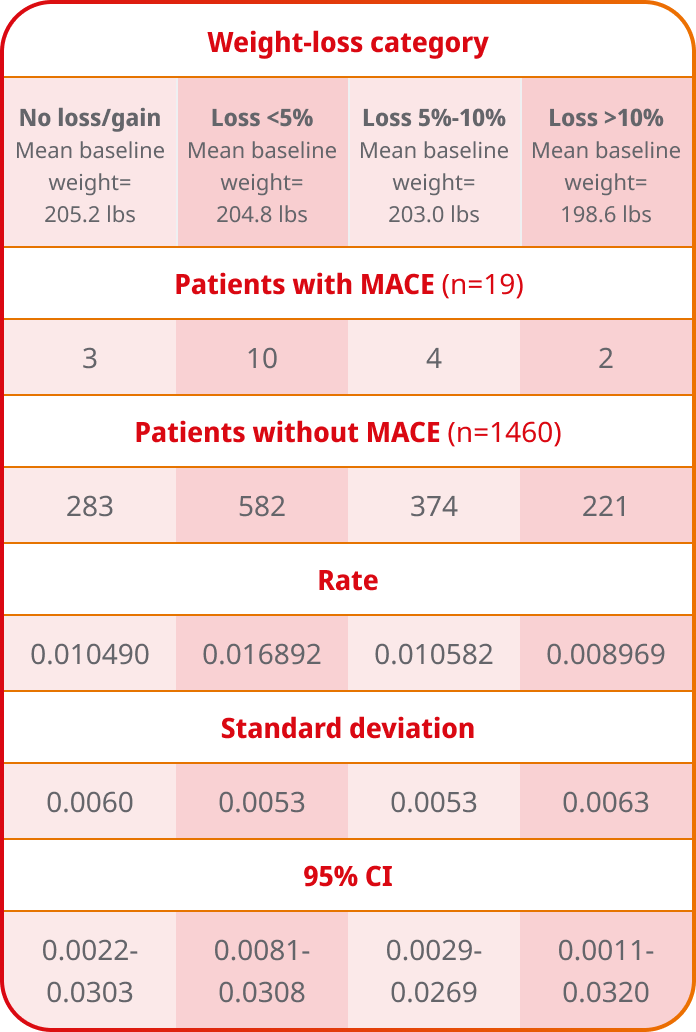

SUSTAIN 6: Post hoc analysis3
Study design: The primary objective of the observational, post hoc analysis of SUSTAIN 6 was to examine the incidence rate of 3-point major adverse cardiovascular events (MACE) (nonfatal ischemic stroke, nonfatal MI, and CV death) by weight-loss categories during the first 13 months after initiation of Ozempic® injection (N=1479). Weight loss was defined as the difference in weight recorded between Visit 2 (Week 0) and Visit 11 (Week 56). CV events are captured from Visit 11 to Visit 25 (Week 104), with a median follow-up of 2.1 years. Patients experiencing any of the composite 3-point MACE or who had incomplete data (including those who withdrew) during the weight-loss period were excluded from the analysis.
Limitations: The study is subject to the inherent limitations typical of post hoc clinical trial analyses. Sample size (N=1479) and generalizability are the most limiting factors of this analysis as they are limited to the data collected during the SUSTAIN 6 trial and are focused only on patients who were taking Ozempic® injection. Adjustments for potential confounding variables were made, though residual and unmeasured confounding is still a possibility. To better adjust for confounders, specific covariates were selected to model the relationship between confounding factors and the exposure. There are other variables that may influence the relationship between weight-loss category and CV events during the trial, such as family history or length of atherosclerotic CVD history, which we do not have access to in this data set. Additionally, there may have been selection bias in the original recruitment of the trial, which spans across multiple countries.
Ozempic® pen has once-weekly dosing1
More ways to help your patients with T2D
ARR=absolute risk reduction; CI=confidence interval; CV=cardiovascular; CVD=cardiovascular disease; CVOT=cardiovascular outcomes trial; HR=hazard ratio; MACE=major adverse cardiovascular event; MI=myocardial infarction; NNT=number needed to treat; NS=not significant; OAD=oral antidiabetic drug; RRR=relative risk reduction; SOC=standard of care; T2D=type 2 diabetes.
Important Safety Information for Ozempic®
WARNING: RISK OF THYROID C-CELL TUMORS
- In rodents, semaglutide causes dose-dependent and treatment-duration- dependent thyroid C-cell tumors at clinically relevant exposures. It is unknown whether Ozempic® causes thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans as human relevance of semaglutide-induced rodent thyroid C-cell tumors has not been determined
- Ozempic® is contraindicated in patients with a personal or family history of MTC or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Counsel patients regarding the potential risk for MTC with the use of Ozempic® and inform them of symptoms of thyroid tumors (e.g., a mass in the neck, dysphagia, dyspnea, persistent hoarseness). Routine monitoring of serum calcitonin or using thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with Ozempic®
Contraindications
- Ozempic® is contraindicated in patients with a personal or family history of MTC or in patients with MEN 2, and in patients with a prior serious hypersensitivity reaction to semaglutide or to any of the excipients in Ozempic®. Serious hypersensitivity reactions including anaphylaxis and angioedema have been reported with Ozempic®
Warnings and Precautions
- Risk of Thyroid C-Cell Tumors: Patients should be further evaluated if serum calcitonin is measured and found to be elevated or thyroid nodules are noted on physical examination or neck imaging
- Acute Pancreatitis: Acute pancreatitis, including fatal and nonfatal hemorrhagic or necrotizing pancreatitis, has been observed in patients treated with GLP-1 receptor agonists, including semaglutide. Observe patients carefully for signs and symptoms of acute pancreatitis, which may include persistent or severe abdominal pain (sometimes radiating to the back), with or without nausea or vomiting. If pancreatitis is suspected, discontinue Ozempic® and initiate appropriate management
- Diabetic Retinopathy Complications: In a 2-year trial involving patients with type 2 diabetes and high cardiovascular risk, more events of diabetic retinopathy complications occurred in patients treated with Ozempic® injection (3%) compared to placebo (1.8%). In a pooled analysis of glycemic control trials, patients reported diabetic retinopathy related adverse reactions during the trial (4.2% with semaglutide tablets and 3.8% with comparator). The absolute risk increase for diabetic retinopathy complications was larger among patients with a history of diabetic retinopathy at baseline than among patients without a known history.
Rapid improvement in glucose control has been associated with a temporary worsening of diabetic retinopathy. Patients with a history of diabetic retinopathy should be monitored for progression of diabetic retinopathy - Never Share an Ozempic® Pen Between Patients: Ozempic® pens must never be shared between patients, even if the needle is changed. Pen-sharing poses a risk for transmission of blood-borne pathogens
- Hypoglycemia: Patients receiving Ozempic® in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin may have an increased risk of hypoglycemia, including severe hypoglycemia. Inform patients using these concomitant medications of the risk of hypoglycemia and educate them on the signs and symptoms of hypoglycemia
- Acute Kidney Injury Due to Volume Depletion: There have been postmarketing reports of acute kidney injury, in some cases requiring hemodialysis, in patients treated with semaglutide. The majority of reported events occurred in patients who experienced gastrointestinal reactions leading to dehydration such as nausea, vomiting, or diarrhea. Monitor renal function in patients reporting adverse reactions to Ozempic® that could lead to volume depletion, especially during dosage initiation and escalation
- Severe Gastrointestinal (GI) Adverse Reactions: Use of Ozempic® has been associated with GI adverse reactions, sometimes severe. In clinical trials, severe GI adverse reactions were reported more frequently among patients who received Ozempic® injection (0.5 mg 0.4%, 1 mg 0.8%) than placebo (0%); and severe GI adverse reactions were reported more frequently among patients who received semaglutide tablets (7 mg 0.6%, 14 mg 2%) than placebo (0.3%). Severe GI adverse reactions have also been reported postmarketing with GLP-1 receptor agonists. Ozempic® is not recommended in patients with severe gastroparesis
- Hypersensitivity: Serious hypersensitivity reactions (e.g., anaphylaxis, angioedema) have been reported in patients treated with Ozempic®. If hypersensitivity reactions occur, discontinue use of Ozempic®; treat promptly per standard of care, and monitor until signs and symptoms resolve. Use caution in a patient with a history of angioedema or anaphylaxis with another GLP-1 receptor agonist
- Acute Gallbladder Disease: Acute events of gallbladder disease such as cholelithiasis or cholecystitis have been reported in GLP-1 receptor agonist trials and postmarketing. In placebo-controlled trials, cholelithiasis was reported in 1.5% and 0.4% of patients treated with Ozempic® injection 0.5 mg and 1 mg, respectively, and not reported in placebo-treated patients. In placebo-controlled trials to improve glycemic control, cholelithiasis was reported in 1% of patients treated with semaglutide tablets 7 mg. In a 4-year CV outcomes trial (Trial 7), cholelithiasis was reported in 1.1% of patients treated with semaglutide tablets 14 mg and in 0.9% of placebo-treated patients. In Trial 7, cholecystitis was reported in 1.1% treated with semaglutide tablets 14 mg and in 0.7% of placebo-treated patients. If cholelithiasis is suspected, gallbladder studies and appropriate clinical follow-up are indicated
- Pulmonary Aspiration During General Anesthesia or Deep Sedation: Ozempic® delays gastric emptying. There have been rare postmarketing reports of pulmonary aspiration in patients receiving GLP-1 receptor agonists undergoing elective surgeries or procedures requiring general anesthesia or deep sedation who had residual gastric contents despite reported adherence to preoperative fasting recommendations. Instruct patients to inform healthcare providers prior to any planned surgeries or procedures if they are taking Ozempic®
Adverse Reactions
- The most common adverse reactions reported in ≥5% of patients taking Ozempic® are nausea, vomiting, diarrhea, abdominal pain, and constipation. Decreased appetite was also reported in ≥5% of patients taking semaglutide tablets
Drug Interactions
- When initiating Ozempic®, consider reducing the dose of concomitantly administered insulin secretagogue (such as sulfonylureas) or insulin to reduce the risk of hypoglycemia
- Ozempic® causes a delay of gastric emptying and has the potential to impact the absorption of concomitantly administered oral medications. Monitor the effects of oral medications concomitantly administered with Ozempic®. Consider increased clinical or laboratory monitoring for medications that have a narrow therapeutic index or that require clinical monitoring
Use in Specific Populations
- Pregnancy: Available data with semaglutide use in pregnant women are not sufficient to determine a drug associated risk for major birth defects, miscarriage, or other adverse maternal or fetal outcomes. Discontinue Ozempic® in women at least 2 months before a planned pregnancy due to the long washout period for semaglutide
- Lactation: A clinical lactation study reported semaglutide concentrations below the lower limit of quantification in human breast milk. However, salcaprozate sodium (SNAC) and/or its metabolites are present in human milk. Because of the unknown potential for serious adverse reactions in the breastfed infant due to the possible accumulation of SNAC, an absorption enhancer for Ozempic® tablets, and because there are alternative formulations of semaglutide that do not contain SNAC that can be used during lactation, advise patients that breastfeeding is not recommended during treatment with Ozempic® tablets
Please click here for Ozempic® injection Prescribing Information, including Boxed Warning.
Please click here for Ozempic® tablets Prescribing Information, including Boxed Warning.
Indications and Usage
Ozempic® (semaglutide) injection 0.5 mg, 1 mg, or 2 mg and Ozempic® (semaglutide) tablets 4 mg or 9 mg are indicated:
- as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes
Ozempic® injection is indicated:
- to reduce the risk of major adverse cardiovascular (CV) events (CV death, nonfatal myocardial infarction, or nonfatal stroke) in adults with type 2 diabetes and established CV disease
- to reduce the risk of sustained eGFR decline, end-stage kidney disease, and cardiovascular death in adults with type 2 diabetes and chronic kidney disease
Ozempic® tablets are indicated:
- to reduce the risk of major adverse cardiovascular (CV) events (CV death, nonfatal myocardial infarction, or nonfatal stroke) in adults with type 2 diabetes who are at high risk for these events
Important Safety Information for Ozempic®
WARNING: RISK OF THYROID C-CELL TUMORS
- In rodents, semaglutide causes dose-dependent and treatment-duration- dependent thyroid C-cell tumors at clinically relevant exposures. It is unknown whether Ozempic® causes thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans as human relevance of semaglutide-induced rodent thyroid C-cell tumors has not been determined
- Ozempic® is contraindicated in patients with a personal or family history of MTC or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Counsel patients regarding the potential risk for MTC with the use of Ozempic® and inform them of symptoms of thyroid tumors (e.g., a mass in the neck, dysphagia, dyspnea, persistent hoarseness). Routine monitoring of serum calcitonin or using thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with Ozempic®
Important Safety Information for Ozempic®
WARNING: RISK OF THYROID C-CELL TUMORS
- In rodents, semaglutide causes dose-dependent and treatment-duration- dependent thyroid C-cell tumors at clinically relevant exposures. It is unknown whether Ozempic® causes thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans as human relevance of semaglutide-induced rodent thyroid C-cell tumors has not been determined
- Ozempic® is contraindicated in patients with a personal or family history of MTC or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Counsel patients regarding the potential risk for MTC with the use of Ozempic® and inform them of symptoms of thyroid tumors (e.g., a mass in the neck, dysphagia, dyspnea, persistent hoarseness). Routine monitoring of serum calcitonin or using thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with Ozempic®
Contraindications
- Ozempic® is contraindicated in patients with a personal or family history of MTC or in patients with MEN 2, and in patients with a prior serious hypersensitivity reaction to semaglutide or to any of the excipients in Ozempic®. Serious hypersensitivity reactions including anaphylaxis and angioedema have been reported with Ozempic®
Warnings and Precautions
- Risk of Thyroid C-Cell Tumors: Patients should be further evaluated if serum calcitonin is measured and found to be elevated or thyroid nodules are noted on physical examination or neck imaging
- Acute Pancreatitis: Acute pancreatitis, including fatal and nonfatal hemorrhagic or necrotizing pancreatitis, has been observed in patients treated with GLP-1 receptor agonists, including semaglutide. Observe patients carefully for signs and symptoms of acute pancreatitis, which may include persistent or severe abdominal pain (sometimes radiating to the back), with or without nausea or vomiting. If pancreatitis is suspected, discontinue Ozempic® and initiate appropriate management
- Diabetic Retinopathy Complications: In a 2-year trial involving patients with type 2 diabetes and high cardiovascular risk, more events of diabetic retinopathy complications occurred in patients treated with Ozempic® injection (3%) compared to placebo (1.8%). In a pooled analysis of glycemic control trials, patients reported diabetic retinopathy related adverse reactions during the trial (4.2% with semaglutide tablets and 3.8% with comparator). The absolute risk increase for diabetic retinopathy complications was larger among patients with a history of diabetic retinopathy at baseline than among patients without a known history.
Rapid improvement in glucose control has been associated with a temporary worsening of diabetic retinopathy. Patients with a history of diabetic retinopathy should be monitored for progression of diabetic retinopathy - Never Share an Ozempic® Pen Between Patients: Ozempic® pens must never be shared between patients, even if the needle is changed. Pen-sharing poses a risk for transmission of blood-borne pathogens
- Hypoglycemia: Patients receiving Ozempic® in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin may have an increased risk of hypoglycemia, including severe hypoglycemia. Inform patients using these concomitant medications of the risk of hypoglycemia and educate them on the signs and symptoms of hypoglycemia
- Acute Kidney Injury Due to Volume Depletion: There have been postmarketing reports of acute kidney injury, in some cases requiring hemodialysis, in patients treated with semaglutide. The majority of reported events occurred in patients who experienced gastrointestinal reactions leading to dehydration such as nausea, vomiting, or diarrhea. Monitor renal function in patients reporting adverse reactions to Ozempic® that could lead to volume depletion, especially during dosage initiation and escalation
- Severe Gastrointestinal (GI) Adverse Reactions: Use of Ozempic® has been associated with GI adverse reactions, sometimes severe. In clinical trials, severe GI adverse reactions were reported more frequently among patients who received Ozempic® injection (0.5 mg 0.4%, 1 mg 0.8%) than placebo (0%); and severe GI adverse reactions were reported more frequently among patients who received semaglutide tablets (7 mg 0.6%, 14 mg 2%) than placebo (0.3%). Severe GI adverse reactions have also been reported postmarketing with GLP-1 receptor agonists. Ozempic® is not recommended in patients with severe gastroparesis
- Hypersensitivity: Serious hypersensitivity reactions (e.g., anaphylaxis, angioedema) have been reported in patients treated with Ozempic®. If hypersensitivity reactions occur, discontinue use of Ozempic®; treat promptly per standard of care, and monitor until signs and symptoms resolve. Use caution in a patient with a history of angioedema or anaphylaxis with another GLP-1 receptor agonist
- Acute Gallbladder Disease: Acute events of gallbladder disease such as cholelithiasis or cholecystitis have been reported in GLP-1 receptor agonist trials and postmarketing. In placebo-controlled trials, cholelithiasis was reported in 1.5% and 0.4% of patients treated with Ozempic® injection 0.5 mg and 1 mg, respectively, and not reported in placebo-treated patients. In placebo-controlled trials to improve glycemic control, cholelithiasis was reported in 1% of patients treated with semaglutide tablets 7 mg. In a 4-year CV outcomes trial (Trial 7), cholelithiasis was reported in 1.1% of patients treated with semaglutide tablets 14 mg and in 0.9% of placebo-treated patients. In Trial 7, cholecystitis was reported in 1.1% treated with semaglutide tablets 14 mg and in 0.7% of placebo-treated patients. If cholelithiasis is suspected, gallbladder studies and appropriate clinical follow-up are indicated
- Pulmonary Aspiration During General Anesthesia or Deep Sedation: Ozempic® delays gastric emptying. There have been rare postmarketing reports of pulmonary aspiration in patients receiving GLP-1 receptor agonists undergoing elective surgeries or procedures requiring general anesthesia or deep sedation who had residual gastric contents despite reported adherence to preoperative fasting recommendations. Instruct patients to inform healthcare providers prior to any planned surgeries or procedures if they are taking Ozempic®
Adverse Reactions
- The most common adverse reactions reported in ≥5% of patients taking Ozempic® are nausea, vomiting, diarrhea, abdominal pain, and constipation. Decreased appetite was also reported in ≥5% of patients taking semaglutide tablets
Drug Interactions
- When initiating Ozempic®, consider reducing the dose of concomitantly administered insulin secretagogue (such as sulfonylureas) or insulin to reduce the risk of hypoglycemia
- Ozempic® causes a delay of gastric emptying and has the potential to impact the absorption of concomitantly administered oral medications. Monitor the effects of oral medications concomitantly administered with Ozempic®. Consider increased clinical or laboratory monitoring for medications that have a narrow therapeutic index or that require clinical monitoring
Use in Specific Populations
- Pregnancy: Available data with semaglutide use in pregnant women are not sufficient to determine a drug associated risk for major birth defects, miscarriage, or other adverse maternal or fetal outcomes. Discontinue Ozempic® in women at least 2 months before a planned pregnancy due to the long washout period for semaglutide
- Lactation: A clinical lactation study reported semaglutide concentrations below the lower limit of quantification in human breast milk. However, salcaprozate sodium (SNAC) and/or its metabolites are present in human milk. Because of the unknown potential for serious adverse reactions in the breastfed infant due to the possible accumulation of SNAC, an absorption enhancer for Ozempic® tablets, and because there are alternative formulations of semaglutide that do not contain SNAC that can be used during lactation, advise patients that breastfeeding is not recommended during treatment with Ozempic® tablets
Please click here for Ozempic® injection Prescribing Information, including Boxed Warning.
Please click here for Ozempic® tablets Prescribing Information, including Boxed Warning.
Indications and Usage
Ozempic® (semaglutide) injection 0.5 mg, 1 mg, or 2 mg and Ozempic® (semaglutide) tablets 4 mg or 9 mg are indicated:
- as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes
Ozempic® injection is indicated:
- to reduce the risk of major adverse cardiovascular (CV) events (CV death, nonfatal myocardial infarction, or nonfatal stroke) in adults with type 2 diabetes and established CV disease
- to reduce the risk of sustained eGFR decline, end-stage kidney disease, and cardiovascular death in adults with type 2 diabetes and chronic kidney disease
Ozempic® tablets are indicated:
- to reduce the risk of major adverse cardiovascular (CV) events (CV death, nonfatal myocardial infarction, or nonfatal stroke) in adults with type 2 diabetes who are at high risk for these events
References:
- Ozempic injection. Prescribing information. Novo Nordisk Inc.
- Marso SP, Bain SC, Consoli A, et al; SUSTAIN-6 Investigators. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375(19):1834-1844. doi:10.1056/NEJMoa1607141
- Data on file. Novo Nordisk Inc; Plainsboro, NJ.
- Marso SP, Bain SC, Consoli A, et al; SUSTAIN-6 Investigators. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. Supplementary appendix. N Engl J Med. 2016;375(19):1834-1844. doi:10.1056/NEJMoa1607141
- Das SR, Everett BM, Birtcher KK, et al. 2020 Expert consensus decision pathway on novel therapies for cardiovascular risk reduction in patients with type 2 diabetes: a report of the American College of Cardiology Solution Set Oversight Committee. J Am Coll Cardiol. 2020;76(9):1117-1145. doi:10.1016/j.jacc.2020.05.037
- Ozempic tablets. Prescribing information. Novo Nordisk Inc.
- McGuire DK, Marx N, Mulvagh SL, et al; SOUL Study Group. Oral semaglutide and cardiovascular outcomes in high-risk type 2 diabetes. N Engl J Med. 2025;392(20):2001-2012. doi:10.1056/NEJMoa2501006
- Nielsen MS, Brøndsted L, Kankam M, et al. A bioequivalence study of two formulations of oral semaglutide in healthy participants. Diabetes Ther. 2025;16(2):269-287. doi:10.1007/s13300-024-01674-8
- Blumenthal RS, Morris PB, Gaudino M, et al; Peer Review Committee Members. 2026 ACC/AHA/AACVPR/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Dyslipidemia: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. Published online March 13, 2026. doi: 10.1016/j.jacc.2025.11.016
- McGuire DK, Marx N, Mulvagh SL, et al; SOUL Study Group. Oral semaglutide and cardiovascular outcomes in high-risk type 2 diabetes. Supplementary appendix. N Engl J Med. 2025;392(20):2001-2012. doi:10.1056/NEJMoa2501006