Indicated for use in adults and children with hemophilia A for on-demand treatment and control of bleeding episodes, perioperative management of bleeding, and routine prophylaxis to reduce the frequency of bleeding episodes. Esperoct® is not indicated for the treatment of von Willebrand disease.
Longer half-life compared to SHL FVIII products3,4
In patients ≥18 years of age
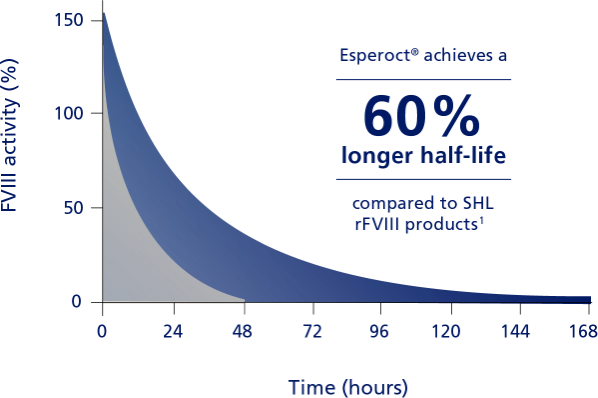
In pathfinder1, a phase 1 study with adult patients, Esperoct® achieved a 60% longer half-life compared to SHL FVIII products.3
Keep them covered longer
Esperoct® achieved a 22-hour half-life in adults.5,a
in adults with site-directed glycoPEGylation4
aIn a phase 3, open-label study, safety, efficacy, and pharmacokinetics (PK) of Esperoct® were evaluated in previously treated patients (PTPs) aged ≥12 years with severe hemophilia A. Single-dose PK studies were performed in 42 adults after receiving Esperoct® 50 IU/kg (includes adult patients from pathfinder2 Phase 3 trial and Phase 1 PK trials).5,6
In patients <12 years of age
In pathfinder5, a phase 3 open-label study with pediatric previously treated patients, Esperoct® achieved an 85% longer half-life compared to SHL FVIII products.4
Compared with SHL FVIII products

Keep them covered longer
In pathfinder5, a phase 3 study of children (aged <12 years), Esperoct® achieved a 14.3-hour mean half-life.4,b
mean half-life in children4,b
bFor children aged 6-11 years.
Achieve high trough levels for a broad range of patients
The pharmacokinetics (PK) of Esperoct® were evaluated in previously treated patients with severe hemophilia A. Esperoct® achieved high factor levels in adolescents, adults, and children.5,c
cFor children aged 6-11 years, trough level is 2%.
In patients aged 12 to 70 years
In patients aged 0 to <12 years
Pivotal trial main phase
In the pathfinder2 open-label study, Esperoct® achieved factor levels ≥3% for the entire dosing interval and in PK modeling data ≥5% for 90% of the time.5
6+ years post hoc analysis
In a post hoc analysis, long-term prophylaxis (≥6 years) showed a stabilization in mean FVIII trough levels and decrease in annualized bleed rate over time.7,g
In patients aged 0 to <12 years
Pivotal trial main phase
In the pathfinder5 open-label study, Esperoct® achieved factor levels ≥2% for the entire dosing interval for children aged 6 to 11 years, and in PK modeling data, ≥5% for 72% of the dosing interval for children aged 0 to <12 years.5
Aged 6-11 years
Aged 0 to <12 years
5+ years post hoc analysis
In a post hoc analysis, long-term prophylaxis (≥5 years) showed a stabilization in mean FVIII trough levels.7,h
dMean trough levels for adolescents (aged 12-<18 years) were 2.7 IU/dL.
eSteady-state FVIII activity profiles were estimated in 143 patients using a one-compartment model with first-order elimination with PK parameters of clearance and volume of distribution.5
fSteady-state FVIII activity profiles were estimated in 22 children using a one-compartment model with first-order elimination, including PK parameters of clearance and volume of distribution.5
gPost hoc analyses were performed on data from the pathfinder2 trial of patients aged ≥12 years with severe hemophilia A who received prophylaxis Q4D. Exploratory descriptive analyses of the data were used to evaluate long-term annual bleed rates and mean factor VIII trough levels which were assessed over time in 53 patients who received ≥6 years of prophylaxis, every 4 days. Limitations of the analyses include the lack of baseline joint status data, which is clinically relevant for phenotypic assessment prior to treatment initiation. The absence limits the ability to draw conclusions regarding improvement in joint status over time. Several trough-level data were excluded if it was believed that they were elevated due to dosing to treat a recent bleed.7
hPost hoc analyses were performed on data from the pathfinder5 trial of patients (aged <12 years) with severe hemophilia A. Exploratory descriptive analyses of the data were used to evaluate mean factor VIII trough levels which were assessed over time in 54 patients who received ≥5 years of twice-weekly prophylaxis. Limitations of the analyses include the exclusion of several trough-level data if believed that they were elevated due to dosing to treat a recent bleed.7
Study designs
Tiede et al (2013)3
pathfinder1 phase 1 clinical trial
Patients: Previously treated males aged ≥18 years (n=26) with severe hemophilia A, a residual FVIII activity of <1%, and at least 150 days of exposure to an FVIII product.
Study design: Open-label dose-escalation trial. Patients received a single dose of 25, 50, or 75 IU/kg of their previous SHL product (pdFVIII or rFVIII), followed by the same dose of Esperoct® after a minimum 4-day wash-out period. To allow for comparison, all results were adjusted to a 50 IU/kg dose of each product.3
Primary endpoint: Evaluating adverse events and antibody formation, including inhibitors against FVIII and Esperoct®, and binding antibodies against Esperoct®.
Secondary endpoints: Evaluating the PK profile of Esperoct® and comparing Esperoct® FVIII activity with that of the patients’ previous FVIII products.3
Giangrande et al (2017 and 2020)1,6
pathfinder2 phase 3 clinical trial: main phase and extension
Patients: Previously treated males aged 12–66 years with severe congenital hemophilia A (FVIII activity <1%) and at least 150 exposure days to any FVIII products.6
Study design: Multinational, open-label, non-randomized trial evaluating safety, pharmacokinetics, and clinical efficacy of Esperoct® when used for prophylaxis and treatment of bleeds. During the main phase, 175 patients received routine prophylaxis (50 IU/kg every 4 days), and 12 adults elected to be treated on-demand. A subset of patients (n=150) continued on in extension phase part 1; 139 continued into the non-randomized extension part 2, with 113 completing the trial.1,6
Co-primary endpoints: Incidence of FVIII inhibitors ≥0.6 BU (Bethesda units) and ABR (annualized bleeding rate) for patients on prophylaxis.6
Meunier et al (2017)4
pathfinder5 phase 3 clinical trial
Patients: Previously treated males (n=68) aged <12 years with severe hemophilia A (FVIII activity level of <1%) and a body weight ≥10 kg. Patients aged 0–5 years had at least 50 exposure days to other FVIII products. Patients aged 6–11 years had at least 150 exposure days.
Study design: Multinational, open-label, non-randomized, non-controlled trial evaluating the safety, clinical efficacy, and pharmacokinetics of prophylactic and treatment on demand of Esperoct®. For bleeding episodes, treatment dose was 20–75 IU/kg, depending on bleed severity and location. For prophylaxis, patients administered 60 IU/kg (50–75 IU/kg) twice weekly for 26 weeks.
Prior to the start of routine prophylaxis, single-dose PK comparison was performed in 27 children between previous SHL products and Esperoct® at the same administered dose. Half-life comparison is based on estimated terminal half-life ratio using a population-based method.
Primary endpoint: Incidence of inhibitory antibodies against FVIII, indicated by a result of ≥0.6 BU (Bethesda units), as measured with the Nijmegen-modified Bethesda assay with heat inactivation of residual FVIII activity.
Secondary endpoint: Frequency of adverse events, including serious adverse effects, inhibitor formation against FVIII, allergic reactions, thromboembolic events, and medication errors.
One proven starting dose for routine prophylaxis (adults and adolescents)
- No dose adjustment needed or related PK testing required5,6
- Regimen can be individualized to less or more frequent dosing based on bleeding episodes5
PEGylation technology
Esperoct® extends half-life through PEGylation.3,5
Important Safety Information for Esperoct®
Contraindications
- Do not use in patients who have known hypersensitivity to Esperoct® or its components, including hamster proteins
Warnings and Precautions
- Hypersensitivity reactions, including anaphylaxis, may occur. Should hypersensitivity reactions occur, discontinue Esperoct® and administer appropriate treatment
- Development of neutralizing antibodies (inhibitors) has occurred. Perform an assay that measures Factor VIII inhibitor concentration if bleeding is not controlled with the recommended dose of Esperoct® or if the expected plasma Factor VIII activity levels are not attained
- Temporary decrease in Factor VIII incremental recovery (IR) has been observed after Esperoct® infusion, within the first 5 exposure days, in previously untreated patients (PUPs) <6 years of age. During the decreased IR period, these subjects may have an increased bleeding tendency. If bleeding is not controlled with the recommended dose of Esperoct® and/or the expected Factor VIII activity levels are not attained and Factor VIII inhibitors are not detected, consider adjusting the dose, dosing frequency, or discontinuing Esperoct®
Adverse Reactions
- The most frequently reported adverse reactions in clinical trials (≥1%) were rash, redness, itching (pruritus), and injection site reactions. Additional frequently reported adverse reactions (≥1%) in PUPs included Factor VIII inhibition and hypersensitivity.
Please click here for Esperoct® Prescribing Information.
Indications and Usage
Esperoct® [antihemophilic factor (recombinant), glycopegylated-exei] is indicated for use in adults and children with hemophilia A for on-demand treatment and control of bleeding episodes, perioperative management of bleeding, and routine prophylaxis to reduce the frequency of bleeding episodes
- Esperoct® is not indicated for the treatment of von Willebrand disease
Important Safety Information for Esperoct®
Contraindications
- Do not use in patients who have known hypersensitivity to Esperoct® or its components, including hamster proteins
Warnings and Precautions
- Hypersensitivity reactions, including anaphylaxis, may occur. Should hypersensitivity reactions occur, discontinue Esperoct® and administer appropriate treatment
- Development of neutralizing antibodies (inhibitors) has occurred. Perform an assay that measures Factor VIII inhibitor concentration if bleeding is not controlled with the recommended dose of Esperoct® or if the expected plasma Factor VIII activity levels are not attained
- Temporary decrease in Factor VIII incremental recovery (IR) has been observed after Esperoct® infusion, within the first 5 exposure days, in previously untreated patients (PUPs) <6 years of age. During the decreased IR period, these subjects may have an increased bleeding tendency. If bleeding is not controlled with the recommended dose of Esperoct® and/or the expected Factor VIII activity levels are not attained and Factor VIII inhibitors are not detected, consider adjusting the dose, dosing frequency, or discontinuing Esperoct®
Adverse Reactions
- The most frequently reported adverse reactions in clinical trials (≥1%) were rash, redness, itching (pruritus), and injection site reactions. Additional frequently reported adverse reactions (≥1%) in PUPs included Factor VIII inhibition and hypersensitivity.
Please click here for Esperoct® Prescribing Information.
Indications and Usage
Esperoct® [antihemophilic factor (recombinant), glycopegylated-exei] is indicated for use in adults and children with hemophilia A for on-demand treatment and control of bleeding episodes, perioperative management of bleeding, and routine prophylaxis to reduce the frequency of bleeding episodes
- Esperoct® is not indicated for the treatment of von Willebrand disease
- Giangrande P, Karim F, Nemes L, et al. Long-term safety and efficacy of N8-GP in previously treated adults and adolescents with hemophilia A: final results from pathfinder2. J Thromb Haemost. 2020;18(1):5-14.
- Šaulytė Trakymiene S, Economou M, Kenet G, Landorph A, Shen C, Kearney S. Long-term safety and efficacy of N8-GP in previously treated pediatric patients with hemophilia A: final results from pathfinder5. J Thromb Haemost. 2020;18(suppl 1):15-25.
- Tiede A, Brand B, Fischer R, et al. Enhancing the pharmacokinetic properties of recombinant factor VIII: first-in-human trial of glycoPEGylated recombinant factor VIII in patients with hemophilia A. J Thromb Haemost. 2013;11(4):670-678.
- Meunier S, Alamelu J, Ehrenforth S, et al. Safety and efficacy of a glycoPEGylated rFVIII (turoctocog alpha pegol, N8-GP) in paediatric patients with severe haemophilia A. Thromb Haemost. 2017;117:1705-1713.
- Esperoct® [package insert]. Plainsboro, NJ: Novo Nordisk Inc.
- Giangrande P, Andreeva T, Chowdary P, et al. Clinical evaluation of glycoPEGylated recombinant FVIII: efficacy and safety in severe haemophilia A. Thromb Haemost. 2017;117(2):252-261.
- Tiede A, Hampton K, Jimenez-Yuste V, Young G, Benchikh El Fegoun S, Chowdry P. Post-hoc analysis on the long-term response to fixed-dose prophylaxis with N8-GP in patients with haemophilia A. Haemophilia. 2021;10.1111/hae.14409.

