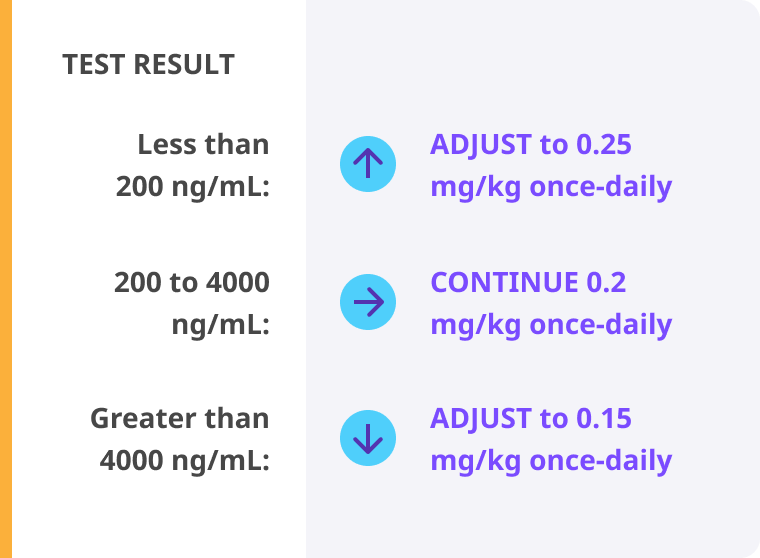
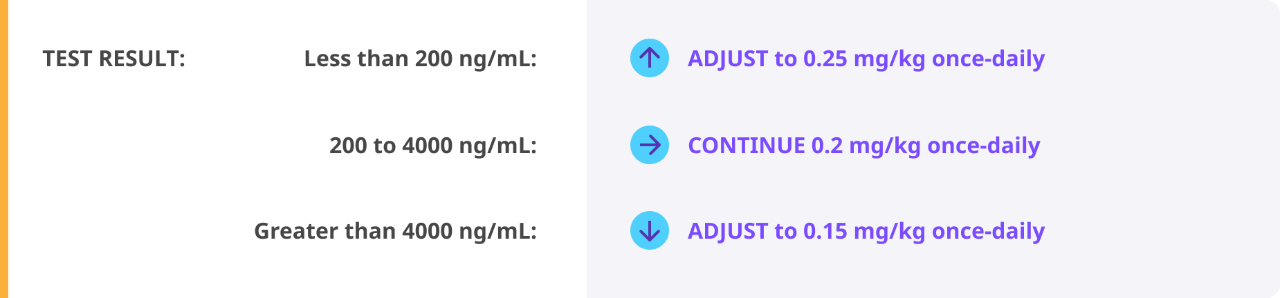
Routine prophylaxis treatment in a prefilled, subcutaneous pen to prevent or reduce the frequency of bleeding episodes in adult and pediatric patients 12 years of age and older who have hemophilia B and A with or without inhibitors
Administration designed with patients in mind
Discover how a daily injection of Alhemo® can fit into your patient’s routine.
Help your patients navigate coverage
NovoCare® is designed for your patients. Patients can access cost savings and support programs.
Important Safety Information for Alhemo®
Contraindications
- Alhemo® is contraindicated in patients with a history of known serious hypersensitivity to Alhemo® or its ingredients
Warnings and Precautions
- Thromboembolic Events (TEs): Venous and arterial TEs were reported in 1.9% of patients (6/320) who also had multiple risk factors, including the use of high doses or prolonged treatment with factor product or bypassing agent (2 of 6 patients). Risk factors for TEs may also include conditions in which tissue factor is overexpressed (eg, atherosclerotic disease, crush injury, cancer, disseminated intravascular coagulation, thrombotic microangiopathy, or septicemia). Inform patients about and monitor them for signs and symptoms of TEs. In case of suspicion of TEs, discontinue Alhemo® and initiate further investigations and management strategies
- Hypersensitivity Reactions: Alhemo® is contraindicated in patients with a history of known serious hypersensitivity to Alhemo® or its ingredients. Hypersensitivity reactions, including erythema, rash, pruritus, and abdominal pain, have occurred in patients treated with Alhemo®. One patient (<1%) experienced anaphylaxis, which resolved after treatment with antihistamines and corticosteroids. Instruct patients of the signs of acute hypersensitivity reactions and to contact their healthcare provider for mild reactions and to seek urgent medical attention for moderate to severe reactions. Discontinue Alhemo® if severe hypersensitivity symptoms occur and initiate medical management
- Increased Laboratory Values of Fibrin D-dimer and Prothrombin Fragment 1.2: Increased levels of fibrin D-dimer and prothrombin fragment 1.2 were seen in 29 (9.1%) and 26 (8.1%) patients, respectively, which is positively correlated with the plasma concentration of concizumab-mtci, indicating a hemostatic effect. For patients taking Alhemo®, these coagulation biomarkers may not be reliable predictive markers for clinical decision-making with suspicion of thrombosis, such as deep vein thrombosis and pulmonary embolism
Adverse Reactions
- The most frequently reported adverse reactions (≥5%) were injection site reactions, headache, and urticaria
- Serious adverse reactions were reported in 6.1% of patients with inhibitors who received Alhemo®. Permanent discontinuation of Alhemo® occurred in 1 patient due to a renal infarct and dosage interruptions of Alhemo® occurred in 1 patient (3%) and was a hypersensitivity reaction
Drug Interactions
- Breakthrough Bleeding Treatment: Take appropriate precautions when treating breakthrough bleeding events in patients receiving Alhemo® prophylaxis and FVIII or FIX or a bypassing agent (eg, rFVIIa or aPCC). For mild and moderate bleeds, the lowest approved dose in the approved product labeling is recommended. For aPCC, a maximum dose of 100 units/kg within 24 hours is recommended. For severe bleeds, follow the dosing instructions in the approved labeling based on clinical judgment
Please click here for Alhemo® Prescribing Information.
Indications and Usage
Alhemo® (concizumab-mtci) injection 60 mg, 150 mg, or 300 mg is indicated for routine prophylaxis to prevent or reduce the frequency of bleeding episodes in adult and pediatric patients 12 years of age and older with hemophilia A or B with or without Factor VIII or IX inhibitors.
Important Safety Information for Alhemo®
Contraindications
- Alhemo® is contraindicated in patients with a history of known serious hypersensitivity to Alhemo® or its ingredients
Warnings and Precautions
- Thromboembolic Events (TEs): Venous and arterial TEs were reported in 1.9% of patients (6/320) who also had multiple risk factors, including the use of high doses or prolonged treatment with factor product or bypassing agent (2 of 6 patients). Risk factors for TEs may also include conditions in which tissue factor is overexpressed (eg, atherosclerotic disease, crush injury, cancer, disseminated intravascular coagulation, thrombotic microangiopathy, or septicemia). Inform patients about and monitor them for signs and symptoms of TEs. In case of suspicion of TEs, discontinue Alhemo® and initiate further investigations and management strategies
- Hypersensitivity Reactions: Alhemo® is contraindicated in patients with a history of known serious hypersensitivity to Alhemo® or its ingredients. Hypersensitivity reactions, including erythema, rash, pruritus, and abdominal pain, have occurred in patients treated with Alhemo®. One patient (<1%) experienced anaphylaxis, which resolved after treatment with antihistamines and corticosteroids. Instruct patients of the signs of acute hypersensitivity reactions and to contact their healthcare provider for mild reactions and to seek urgent medical attention for moderate to severe reactions. Discontinue Alhemo® if severe hypersensitivity symptoms occur and initiate medical management
- Increased Laboratory Values of Fibrin D-dimer and Prothrombin Fragment 1.2: Increased levels of fibrin D-dimer and prothrombin fragment 1.2 were seen in 29 (9.1%) and 26 (8.1%) patients, respectively, which is positively correlated with the plasma concentration of concizumab-mtci, indicating a hemostatic effect. For patients taking Alhemo®, these coagulation biomarkers may not be reliable predictive markers for clinical decision-making with suspicion of thrombosis, such as deep vein thrombosis and pulmonary embolism
Adverse Reactions
- The most frequently reported adverse reactions (≥5%) were injection site reactions, headache, and urticaria
- Serious adverse reactions were reported in 6.1% of patients with inhibitors who received Alhemo®. Permanent discontinuation of Alhemo® occurred in 1 patient due to a renal infarct and dosage interruptions of Alhemo® occurred in 1 patient (3%) and was a hypersensitivity reaction
Drug Interactions
- Breakthrough Bleeding Treatment: Take appropriate precautions when treating breakthrough bleeding events in patients receiving Alhemo® prophylaxis and FVIII or FIX or a bypassing agent (eg, rFVIIa or aPCC). For mild and moderate bleeds, the lowest approved dose in the approved product labeling is recommended. For aPCC, a maximum dose of 100 units/kg within 24 hours is recommended. For severe bleeds, follow the dosing instructions in the approved labeling based on clinical judgment
Please click here for Alhemo® Prescribing Information.
Indications and Usage
Alhemo® (concizumab-mtci) injection 60 mg, 150 mg, or 300 mg is indicated for routine prophylaxis to prevent or reduce the frequency of bleeding episodes in adult and pediatric patients 12 years of age and older with hemophilia A or B with or without Factor VIII or IX inhibitors.
Reference:
- Alhemo® [package insert]. Plainsboro, NJ: Novo Nordisk Inc.