For pediatric patients with growth failure due to inadequate secretion of endogenous growth hormone (GH) and Prader-Willi Syndrome; short stature associated with Noonan Syndrome, Turner Syndrome, and children born small for gestational age; idiopathic short stature; and for the replacement of endogenous GH in adults with growth hormone deficiency (GHD). Please see full indications.
Norditropin® FlexPro® injection instructions
In a usability study, participants reported the FlexPro® pen to be easy to learn to use.1,a,b

Norditropin® FlexPro® injection instructions
In a usability study, participants reported the FlexPro® pen to be easy to learn to use.1,a,b

aNeedles are sold separately and may require a prescription in some states.
bBased on a human factors study of the safety and usability of the Norditropin® FlexPro® 30 mg pen in 94 participants (children ages 10-17 with growth-related disorders; adults with GHD; HCPs; and caregivers). Participants, excluding inpatient nurses, received training and performed injections using a foam cushion, and then completed a device-specific questionnaire. Participants rated the device a 6.7 out of 7 (range 5 to 7 on a scale of 1 to 7, where 1 means “strongly disagree” and 7 means “strongly agree”) for the statement, "FlexPro® was easy to learn to use.”1
How to inject Norditropin®—at a glance
Norditropin® is available as prefilled FlexPro® pens. The injection process includes 5 steps, from preparing the pen to storing or disposing of it afterward.2,a
See Instructions for Use for complete instructions.
aDo not share your Norditropin® Pen and needles with another person. You may give another person an infection or get an infection from them. Do not use your Pen without proper training from your healthcare provider. Make sure that you are confident in giving an injection with the Pen before you start your treatment. If you are blind or have poor eyesight and cannot read the dose counter on the Pen, do not use this Pen without help. Get help from a person with good eyesight who is trained to use the Pen.2
Step 1:
Step 2:
Step 3:
Step 4:
Step 5:
- Patients should wash hands with soap and water. They should check the name, strength, and colored label on their pen to make sure that it contains Norditropin® in the right strength.2
- Pull off the pen cap. Turn the pen 1 or 2 times to check that the Norditropin® in the pen is clear and colorless. If the Norditropin® looks unclear or cloudy, do not use the pen
- Tear the paper tab off of a new disposable needle. Screw the needle straight onto the pen tightly. Always use a new needle for each injection. This reduces the risk of contamination, infection, leakage of Norditropin®, and blocked needles leading to incorrect dosing.
- Pull off the outer needle cap and the inner needle cap, and throw them both away.2
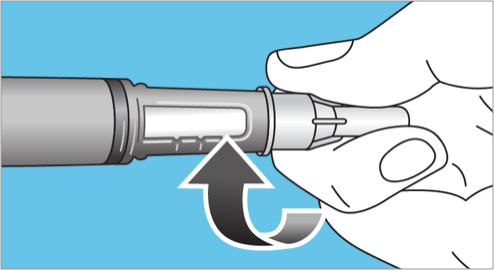
- Before patients inject their first dose using a new FlexPro® pen, patients must check the medication flow by priming the pen2
- Turn the dose selector clockwise to 1 tick mark to select the smallest amount of medicine for the pen
- Hold the Pen with the needle pointing up. Press and hold in the dose button until the dose counter returns to “0” and lines up with the dose pointer. A drop of Norditropin® then appears at the needle tip
- If no drop appears, repeat the priming process up to 6 times. If no drop appears, change the needle and prime one more time. If a drop of liquid still does not appear, call 1-888-NOVO-444 (1-888-668-6444) for help.2
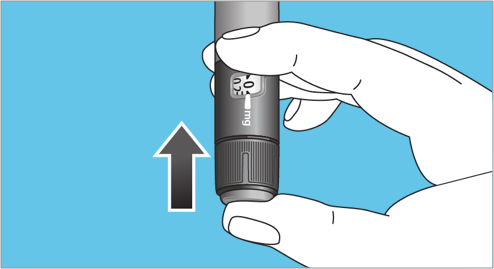
- Select the prescribed dose by turning the dose selector clockwise or counterclockwise until the correct dose in the dose counter lines up with the dose pointer. Do not rely on pen clicks to select a dose
- If there is not enough Norditropin® (somatropin) injection left in the pen to deliver the full prescribed dose, patients may deliver the amount left in the pen. Then, inject the remainder of the dose using a new Norditropin® FlexPro® pen, prepared as described in steps 1 and 2. Patients should only split their dose if they have been trained or advised by their healthcare provider on how to do this2

- Norditropin can be injected under the skin to the back of the upper arm, abdomen, buttocks, or thigh, as instructed by the healthcare provider. It is important to change the injection site every day
- Injection site should be wiped with an alcohol swab and the area should be dry before injecting
- Insert the needle under the skin
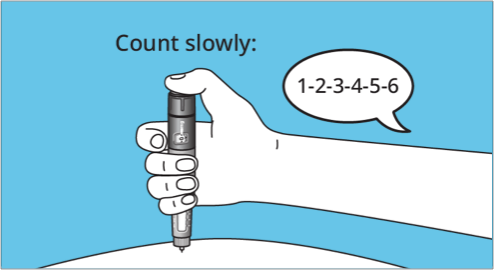
- Press and hold the dose button to inject until the “0” appears in the dose counter and lines up with the dose pointer. A firm click may be heard or felt
- Needle should be kept in the skin after the dose counter has returned to "0." To ensure that the full dose is administered, count slowly to 6
- Carefully remove the needle from the skin. A drop of Norditropin® may appear at the needle tip. This is normal and does not affect the injected amount2
- Patients should carefully remove the needle from the pen by turning the needle counterclockwise and placing it in an appropriate sharps disposal container immediately
- Pen cap should be placed on the pen after each use to protect Norditropin® from light
- When there is not enough Norditropin® in the FlexPro® pen, it may be thrown away in household trash after removal of the needle2

How to store Norditropin®
All unused Norditropin® products must be refrigerated (36 °F to 46 °F) prior to first use. Do not freeze and avoid direct light. After first injection, Norditropin® pens can either be stored outside of the refrigerator (up to 77 °F) for use within 3 weeks or in the refrigerator (between 36 °F and 46 °F) for use within 4 weeks.2

Growth calculators
Our online calculators can help you estimate your patients’ height velocity and other metrics used in clinical practice. For US health care professionals only.
Growth calculators
Our online calculators can help you estimate your patients’ height velocity and other metrics used in clinical practice. For US health care professionals only.
Important Safety Information for Norditropin®
Contraindications
Norditropin® is contraindicated in patients with:
- Acute critical illness after open heart surgery, abdominal surgery or multiple accidental trauma, or those with acute respiratory failure due to the risk of increased mortality with use of pharmacologic doses of somatropin
- Pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to the risk of sudden death
- Active Malignancy
- Hypersensitivity to Norditropin® or any of its excipients. Systemic hypersensitivity reactions have been reported with postmarketing use of somatropins
- Active proliferative or severe non-proliferative diabetic retinopathy
- Pediatric patients with closed epiphyses
Warnings and Precautions
- Increased mortality in patients with acute critical illness due to complications following open heart or abdominal surgery or multiple accidental trauma, or those with respiratory failure has been reported.
- Sudden death in pediatric patients with Prader-Willi Syndrome has been reported after initiating treatment with somatropin with one or more of the following risk factors: severe obesity, history of upper airway obstruction or sleep apnea, or unidentified respiratory infection. Evaluate patients for signs of upper airway obstruction and sleep apnea before initiation of treatment.
- Increased risk of neoplasms: Monitor patients with preexisting tumors for progression or recurrence. In childhood cancer survivors who were treated with radiation to the brain/head for their first neoplasm and who developed subsequent GHD and were treated with somatropin, an increased risk of a second neoplasm, in particular meningiomas, has been reported. Pediatric patients with certain rare genetic causes of short stature have an increased risk of developing malignancies and should be carefully monitored for development of neoplasms. Monitor patients carefully for increased growth, or potential malignant changes of preexisting nevi.
- Glucose intolerance and diabetes mellitus: Treatment with somatropin may decrease insulin sensitivity, particularly at higher doses. New-onset type 2 diabetes mellitus has been reported. Monitor glucose levels in all patients. Doses of concurrent antidiabetic drugs may require adjustment.
- Intracranial hypertension has been reported in a small number of patients, usually within the first 8 weeks of somatropin treatment. Funduscopic examination should be performed before initiating treatment and periodically thereafter.
- Severe hypersensitivity: Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported with postmarketing use of somatropins.
- Fluid retention in adults (clinically manifesting as edema, arthralgia, myalgia, nerve compression syndromes including carpal tunnel syndrome/paraesthesias) may frequently occur and is usually transient and dose dependent.
- Hypoadrenalism: Patients who have or are at risk for pituitary hormone deficiency(s) may be at risk for reduced serum cortisol levels and/or unmasking of central (secondary) hypoadrenalism. In addition, patients treated with glucocorticoid replacement for previously diagnosed hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Norditropin® treatment.
- Hypothyroidism if undiagnosed/untreated, may prevent an optimal response to Norditropin®, in particular, the growth response in pediatric patients. In patients with GHD, central (secondary) hypothyroidism may first become evident or worsen during somatropin treatment. Periodic thyroid function tests and thyroid hormone replacement therapy should be initiated or adjusted when indicated.
- Slipped capital femoral epiphysis in pediatric patients may occur more frequently in patients with endocrine disorders or in patients undergoing rapid growth. Slipped capital femoral epiphysis may lead to osteonecrosis. Cases of slipped capital femoral epiphysis with or without osteonecrosis have been reported in pediatric patients with short stature receiving somatropin, including Norditropin®. Evaluate pediatric patients receiving Norditropin® with the onset of a limp or complaints of hip or knee pain for slipped capital femoral epiphysis and osteonecrosis and manage accordingly.
- Progression of preexisting scoliosis in pediatric patients can occur in patients who experience rapid growth. Patients with a history of scoliosis should be monitored for progression.
- Pancreatitis: Cases of pancreatitis have been reported. Pancreatitis should be considered in any patient who develops persistent severe abdominal pain.
- Lipoatrophy: Tissue atrophy may result when somatropin is administered subcutaneously at the same site over a long period of time. Rotate injection sites when administering Norditropin® to reduce this risk.
Adverse Reactions
- Common adverse reactions in adults and pediatric patients include: upper respiratory infection, fever, pharyngitis, headache, otitis media, edema, arthralgia, paresthesia, myalgia, peripheral edema, flu syndrome, and impaired glucose tolerance
Drug Interactions
- Glucocorticoids: Patients treated with glucocorticoid for hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Norditropin®
- Pharmacologic Glucocorticoid Therapy and Supraphysiologic Glucocorticoid Treatment: Adjust glucocorticoid replacement dosing in pediatric patients receiving glucocorticoid treatment to avoid both hypoadrenalism and an inhibitory effect on growth
- Cytochrome P450-Metabolized Drugs: Norditropin® may alter the clearance. Monitor carefully if used with Norditropin®
- Oral Estrogen: Larger doses of Norditropin® may be required
- Insulin and/or Other Hypoglycemic Agents: Dose adjustment of insulin or hypoglycemic agent may be required
Use in Specific Populations
- Pregnancy and Nursing Mothers: There are limited data with somatropin use in pregnant women and nursing mothers to inform a drug-associated risk for adverse developmental outcomes
- Geriatric Use: The safety and effectiveness in patients aged 65 and over has not been evaluated in clinical studies
Please click here for Norditropin® Prescribing Information.
Indications and Usage
Norditropin® (somatropin) injection 5 mg, 10 mg, or 15 mg is indicated for the treatment of pediatric patients with:
- growth failure due to inadequate secretion of endogenous growth hormone (GH)
- short stature associated with Noonan syndrome
- short stature associated with Turner syndrome
- short stature born small for gestational age (SGA) with no catch-up growth by age 2 to 4 years of age
- Idiopathic Short Stature (ISS), height standard deviation score (SDS) <-2.25, and associated with growth rates unlikely to permit attainment of adult height in the normal range
- growth failure due to Prader-Willi syndrome (PWS)
Norditropin® is also indicated for the replacement of endogenous GH in adults with growth hormone deficiency (GHD)
Indications and Usage
Norditropin® (somatropin) injection 5 mg, 10 mg, or 15 mg is indicated for the treatment of pediatric patients with:
- growth failure due to inadequate secretion of endogenous growth hormone (GH)
- short stature associated with Noonan syndrome
- short stature associated with Turner syndrome
- short stature born small for gestational age (SGA) with no catch-up growth by age 2 to 4 years of age
- Idiopathic Short Stature (ISS), height standard deviation score (SDS) <-2.25, and associated with growth rates unlikely to permit attainment of adult height in the normal range
- growth failure due to Prader-Willi syndrome (PWS)
Norditropin® is also indicated for the replacement of endogenous GH in adults with growth hormone deficiency (GHD)
Important Safety Information for Norditropin®
Contraindications
Norditropin® is contraindicated in patients with:
- Acute critical illness after open heart surgery, abdominal surgery or multiple accidental trauma, or those with acute respiratory failure due to the risk of increased mortality with use of pharmacologic doses of somatropin
- Pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to the risk of sudden death
- Active Malignancy
- Hypersensitivity to Norditropin® or any of its excipients. Systemic hypersensitivity reactions have been reported with postmarketing use of somatropins
- Active proliferative or severe non-proliferative diabetic retinopathy
- Pediatric patients with closed epiphyses
Warnings and Precautions
- Increased mortality in patients with acute critical illness due to complications following open heart or abdominal surgery or multiple accidental trauma, or those with respiratory failure has been reported.
- Sudden death in pediatric patients with Prader-Willi Syndrome has been reported after initiating treatment with somatropin with one or more of the following risk factors: severe obesity, history of upper airway obstruction or sleep apnea, or unidentified respiratory infection. Evaluate patients for signs of upper airway obstruction and sleep apnea before initiation of treatment.
- Increased risk of neoplasms: Monitor patients with preexisting tumors for progression or recurrence. In childhood cancer survivors who were treated with radiation to the brain/head for their first neoplasm and who developed subsequent GHD and were treated with somatropin, an increased risk of a second neoplasm, in particular meningiomas, has been reported. Pediatric patients with certain rare genetic causes of short stature have an increased risk of developing malignancies and should be carefully monitored for development of neoplasms. Monitor patients carefully for increased growth, or potential malignant changes of preexisting nevi.
- Glucose intolerance and diabetes mellitus: Treatment with somatropin may decrease insulin sensitivity, particularly at higher doses. New-onset type 2 diabetes mellitus has been reported. Monitor glucose levels in all patients. Doses of concurrent antidiabetic drugs may require adjustment.
- Intracranial hypertension has been reported in a small number of patients, usually within the first 8 weeks of somatropin treatment. Funduscopic examination should be performed before initiating treatment and periodically thereafter.
- Severe hypersensitivity: Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported with postmarketing use of somatropins.
- Fluid retention in adults (clinically manifesting as edema, arthralgia, myalgia, nerve compression syndromes including carpal tunnel syndrome/paraesthesias) may frequently occur and is usually transient and dose dependent.
- Hypoadrenalism: Patients who have or are at risk for pituitary hormone deficiency(s) may be at risk for reduced serum cortisol levels and/or unmasking of central (secondary) hypoadrenalism. In addition, patients treated with glucocorticoid replacement for previously diagnosed hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Norditropin® treatment.
- Hypothyroidism if undiagnosed/untreated, may prevent an optimal response to Norditropin®, in particular, the growth response in pediatric patients. In patients with GHD, central (secondary) hypothyroidism may first become evident or worsen during somatropin treatment. Periodic thyroid function tests and thyroid hormone replacement therapy should be initiated or adjusted when indicated.
- Slipped capital femoral epiphysis in pediatric patients may occur more frequently in patients with endocrine disorders or in patients undergoing rapid growth. Slipped capital femoral epiphysis may lead to osteonecrosis. Cases of slipped capital femoral epiphysis with or without osteonecrosis have been reported in pediatric patients with short stature receiving somatropin, including Norditropin®. Evaluate pediatric patients receiving Norditropin® with the onset of a limp or complaints of hip or knee pain for slipped capital femoral epiphysis and osteonecrosis and manage accordingly.
- Progression of preexisting scoliosis in pediatric patients can occur in patients who experience rapid growth. Patients with a history of scoliosis should be monitored for progression.
- Pancreatitis: Cases of pancreatitis have been reported. Pancreatitis should be considered in any patient who develops persistent severe abdominal pain.
- Lipoatrophy: Tissue atrophy may result when somatropin is administered subcutaneously at the same site over a long period of time. Rotate injection sites when administering Norditropin® to reduce this risk.
Adverse Reactions
- Common adverse reactions in adults and pediatric patients include: upper respiratory infection, fever, pharyngitis, headache, otitis media, edema, arthralgia, paresthesia, myalgia, peripheral edema, flu syndrome, and impaired glucose tolerance
Drug Interactions
- Glucocorticoids: Patients treated with glucocorticoid for hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Norditropin®
- Pharmacologic Glucocorticoid Therapy and Supraphysiologic Glucocorticoid Treatment: Adjust glucocorticoid replacement dosing in pediatric patients receiving glucocorticoid treatment to avoid both hypoadrenalism and an inhibitory effect on growth
- Cytochrome P450-Metabolized Drugs: Norditropin® may alter the clearance. Monitor carefully if used with Norditropin®
- Oral Estrogen: Larger doses of Norditropin® may be required
- Insulin and/or Other Hypoglycemic Agents: Dose adjustment of insulin or hypoglycemic agent may be required
Use in Specific Populations
- Pregnancy and Nursing Mothers: There are limited data with somatropin use in pregnant women and nursing mothers to inform a drug-associated risk for adverse developmental outcomes
- Geriatric Use: The safety and effectiveness in patients aged 65 and over has not been evaluated in clinical studies
Please click here for Norditropin® Prescribing Information.
Indications and Usage
Norditropin® (somatropin) injection 5 mg, 10 mg, or 15 mg is indicated for the treatment of pediatric patients with:
- growth failure due to inadequate secretion of endogenous growth hormone (GH)
- short stature associated with Noonan syndrome
- short stature associated with Turner syndrome
- short stature born small for gestational age (SGA) with no catch-up growth by age 2 to 4 years of age
- Idiopathic Short Stature (ISS), height standard deviation score (SDS) <-2.25, and associated with growth rates unlikely to permit attainment of adult height in the normal range
- growth failure due to Prader-Willi syndrome (PWS)
Norditropin® is also indicated for the replacement of endogenous GH in adults with growth hormone deficiency (GHD)
Important Safety Information for Norditropin®
Contraindications
Norditropin® is contraindicated in patients with:
- Acute critical illness after open heart surgery, abdominal surgery or multiple accidental trauma, or those with acute respiratory failure due to the risk of increased mortality with use of pharmacologic doses of somatropin
- Pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to the risk of sudden death
- Active Malignancy
- Hypersensitivity to Norditropin® or any of its excipients. Systemic hypersensitivity reactions have been reported with postmarketing use of somatropins
- Active proliferative or severe non-proliferative diabetic retinopathy
- Pediatric patients with closed epiphyses
Important Safety Information for Norditropin®
Contraindications
Norditropin® is contraindicated in patients with:
- Acute critical illness after open heart surgery, abdominal surgery or multiple accidental trauma, or those with acute respiratory failure due to the risk of increased mortality with use of pharmacologic doses of somatropin
- Pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to the risk of sudden death
- Active Malignancy
- Hypersensitivity to Norditropin® or any of its excipients. Systemic hypersensitivity reactions have been reported with postmarketing use of somatropins
- Active proliferative or severe non-proliferative diabetic retinopathy
- Pediatric patients with closed epiphyses
Warnings and Precautions
- Increased mortality in patients with acute critical illness due to complications following open heart or abdominal surgery or multiple accidental trauma, or those with respiratory failure has been reported.
- Sudden death in pediatric patients with Prader-Willi Syndrome has been reported after initiating treatment with somatropin with one or more of the following risk factors: severe obesity, history of upper airway obstruction or sleep apnea, or unidentified respiratory infection. Evaluate patients for signs of upper airway obstruction and sleep apnea before initiation of treatment.
- Increased risk of neoplasms: Monitor patients with preexisting tumors for progression or recurrence. In childhood cancer survivors who were treated with radiation to the brain/head for their first neoplasm and who developed subsequent GHD and were treated with somatropin, an increased risk of a second neoplasm, in particular meningiomas, has been reported. Pediatric patients with certain rare genetic causes of short stature have an increased risk of developing malignancies and should be carefully monitored for development of neoplasms. Monitor patients carefully for increased growth, or potential malignant changes of preexisting nevi.
- Glucose intolerance and diabetes mellitus: Treatment with somatropin may decrease insulin sensitivity, particularly at higher doses. New-onset type 2 diabetes mellitus has been reported. Monitor glucose levels in all patients. Doses of concurrent antidiabetic drugs may require adjustment.
- Intracranial hypertension has been reported in a small number of patients, usually within the first 8 weeks of somatropin treatment. Funduscopic examination should be performed before initiating treatment and periodically thereafter.
- Severe hypersensitivity: Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported with postmarketing use of somatropins.
- Fluid retention in adults (clinically manifesting as edema, arthralgia, myalgia, nerve compression syndromes including carpal tunnel syndrome/paraesthesias) may frequently occur and is usually transient and dose dependent.
- Hypoadrenalism: Patients who have or are at risk for pituitary hormone deficiency(s) may be at risk for reduced serum cortisol levels and/or unmasking of central (secondary) hypoadrenalism. In addition, patients treated with glucocorticoid replacement for previously diagnosed hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Norditropin® treatment.
- Hypothyroidism if undiagnosed/untreated, may prevent an optimal response to Norditropin®, in particular, the growth response in pediatric patients. In patients with GHD, central (secondary) hypothyroidism may first become evident or worsen during somatropin treatment. Periodic thyroid function tests and thyroid hormone replacement therapy should be initiated or adjusted when indicated.
- Slipped capital femoral epiphysis in pediatric patients may occur more frequently in patients with endocrine disorders or in patients undergoing rapid growth. Slipped capital femoral epiphysis may lead to osteonecrosis. Cases of slipped capital femoral epiphysis with or without osteonecrosis have been reported in pediatric patients with short stature receiving somatropin, including Norditropin®. Evaluate pediatric patients receiving Norditropin® with the onset of a limp or complaints of hip or knee pain for slipped capital femoral epiphysis and osteonecrosis and manage accordingly.
- Progression of preexisting scoliosis in pediatric patients can occur in patients who experience rapid growth. Patients with a history of scoliosis should be monitored for progression.
- Pancreatitis: Cases of pancreatitis have been reported. Pancreatitis should be considered in any patient who develops persistent severe abdominal pain.
- Lipoatrophy: Tissue atrophy may result when somatropin is administered subcutaneously at the same site over a long period of time. Rotate injection sites when administering Norditropin® to reduce this risk.
Adverse Reactions
- Common adverse reactions in adults and pediatric patients include: upper respiratory infection, fever, pharyngitis, headache, otitis media, edema, arthralgia, paresthesia, myalgia, peripheral edema, flu syndrome, and impaired glucose tolerance
Drug Interactions
- Glucocorticoids: Patients treated with glucocorticoid for hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Norditropin®
- Pharmacologic Glucocorticoid Therapy and Supraphysiologic Glucocorticoid Treatment: Adjust glucocorticoid replacement dosing in pediatric patients receiving glucocorticoid treatment to avoid both hypoadrenalism and an inhibitory effect on growth
- Cytochrome P450-Metabolized Drugs: Norditropin® may alter the clearance. Monitor carefully if used with Norditropin®
- Oral Estrogen: Larger doses of Norditropin® may be required
- Insulin and/or Other Hypoglycemic Agents: Dose adjustment of insulin or hypoglycemic agent may be required
Use in Specific Populations
- Pregnancy and Nursing Mothers: There are limited data with somatropin use in pregnant women and nursing mothers to inform a drug-associated risk for adverse developmental outcomes
- Geriatric Use: The safety and effectiveness in patients aged 65 and over has not been evaluated in clinical studies
Please click here for Norditropin® Prescribing Information.
Important Safety Information for Norditropin®
Contraindications
Norditropin® is contraindicated in patients with:
- Acute critical illness after open heart surgery, abdominal surgery or multiple accidental trauma, or those with acute respiratory failure due to the risk of increased mortality with use of pharmacologic doses of somatropin
- Pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to the risk of sudden death
- Active Malignancy
- Hypersensitivity to Norditropin® or any of its excipients. Systemic hypersensitivity reactions have been reported with postmarketing use of somatropins
- Active proliferative or severe non-proliferative diabetic retinopathy
- Pediatric patients with closed epiphyses
Warnings and Precautions
- Increased mortality in patients with acute critical illness due to complications following open heart or abdominal surgery or multiple accidental trauma, or those with respiratory failure has been reported.
- Sudden death in pediatric patients with Prader-Willi Syndrome has been reported after initiating treatment with somatropin with one or more of the following risk factors: severe obesity, history of upper airway obstruction or sleep apnea, or unidentified respiratory infection. Evaluate patients for signs of upper airway obstruction and sleep apnea before initiation of treatment.
- Increased risk of neoplasms: Monitor patients with preexisting tumors for progression or recurrence. In childhood cancer survivors who were treated with radiation to the brain/head for their first neoplasm and who developed subsequent GHD and were treated with somatropin, an increased risk of a second neoplasm, in particular meningiomas, has been reported. Pediatric patients with certain rare genetic causes of short stature have an increased risk of developing malignancies and should be carefully monitored for development of neoplasms. Monitor patients carefully for increased growth, or potential malignant changes of preexisting nevi.
- Glucose intolerance and diabetes mellitus: Treatment with somatropin may decrease insulin sensitivity, particularly at higher doses. New-onset type 2 diabetes mellitus has been reported. Monitor glucose levels in all patients. Doses of concurrent antidiabetic drugs may require adjustment.
- Intracranial hypertension has been reported in a small number of patients, usually within the first 8 weeks of somatropin treatment. Funduscopic examination should be performed before initiating treatment and periodically thereafter.
- Severe hypersensitivity: Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported with postmarketing use of somatropins.
- Fluid retention in adults (clinically manifesting as edema, arthralgia, myalgia, nerve compression syndromes including carpal tunnel syndrome/paraesthesias) may frequently occur and is usually transient and dose dependent.
- Hypoadrenalism: Patients who have or are at risk for pituitary hormone deficiency(s) may be at risk for reduced serum cortisol levels and/or unmasking of central (secondary) hypoadrenalism. In addition, patients treated with glucocorticoid replacement for previously diagnosed hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Norditropin® treatment.
- Hypothyroidism if undiagnosed/untreated, may prevent an optimal response to Norditropin®, in particular, the growth response in pediatric patients. In patients with GHD, central (secondary) hypothyroidism may first become evident or worsen during somatropin treatment. Periodic thyroid function tests and thyroid hormone replacement therapy should be initiated or adjusted when indicated.
- Slipped capital femoral epiphysis in pediatric patients may occur more frequently in patients with endocrine disorders or in patients undergoing rapid growth. Slipped capital femoral epiphysis may lead to osteonecrosis. Cases of slipped capital femoral epiphysis with or without osteonecrosis have been reported in pediatric patients with short stature receiving somatropin, including Norditropin®. Evaluate pediatric patients receiving Norditropin® with the onset of a limp or complaints of hip or knee pain for slipped capital femoral epiphysis and osteonecrosis and manage accordingly.
- Progression of preexisting scoliosis in pediatric patients can occur in patients who experience rapid growth. Patients with a history of scoliosis should be monitored for progression.
- Pancreatitis: Cases of pancreatitis have been reported. Pancreatitis should be considered in any patient who develops persistent severe abdominal pain.
- Lipoatrophy: Tissue atrophy may result when somatropin is administered subcutaneously at the same site over a long period of time. Rotate injection sites when administering Norditropin® to reduce this risk.
Adverse Reactions
- Common adverse reactions in adults and pediatric patients include: upper respiratory infection, fever, pharyngitis, headache, otitis media, edema, arthralgia, paresthesia, myalgia, peripheral edema, flu syndrome, and impaired glucose tolerance
Drug Interactions
- Glucocorticoids: Patients treated with glucocorticoid for hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Norditropin®
- Pharmacologic Glucocorticoid Therapy and Supraphysiologic Glucocorticoid Treatment: Adjust glucocorticoid replacement dosing in pediatric patients receiving glucocorticoid treatment to avoid both hypoadrenalism and an inhibitory effect on growth
- Cytochrome P450-Metabolized Drugs: Norditropin® may alter the clearance. Monitor carefully if used with Norditropin®
- Oral Estrogen: Larger doses of Norditropin® may be required
- Insulin and/or Other Hypoglycemic Agents: Dose adjustment of insulin or hypoglycemic agent may be required
Use in Specific Populations
- Pregnancy and Nursing Mothers: There are limited data with somatropin use in pregnant women and nursing mothers to inform a drug-associated risk for adverse developmental outcomes
- Geriatric Use: The safety and effectiveness in patients aged 65 and over has not been evaluated in clinical studies
Please click here for Norditropin® Prescribing Information.
Indications and Usage
Norditropin® (somatropin) injection 5 mg, 10 mg, or 15 mg is indicated for the treatment of pediatric patients with:
- growth failure due to inadequate secretion of endogenous growth hormone (GH)
- short stature associated with Noonan syndrome
- short stature associated with Turner syndrome
- short stature born small for gestational age (SGA) with no catch-up growth by age 2 to 4 years of age
- Idiopathic Short Stature (ISS), height standard deviation score (SDS) <-2.25, and associated with growth rates unlikely to permit attainment of adult height in the normal range
- growth failure due to Prader-Willi syndrome (PWS)
Norditropin® is also indicated for the replacement of endogenous GH in adults with growth hormone deficiency (GHD)
Indications and Usage
Norditropin® (somatropin) injection 5 mg, 10 mg, or 15 mg is indicated for the treatment of pediatric patients with:
- growth failure due to inadequate secretion of endogenous growth hormone (GH)
- short stature associated with Noonan syndrome
- short stature associated with Turner syndrome
- short stature born small for gestational age (SGA) with no catch-up growth by age 2 to 4 years of age
- Idiopathic Short Stature (ISS), height standard deviation score (SDS) <-2.25, and associated with growth rates unlikely to permit attainment of adult height in the normal range
- growth failure due to Prader-Willi syndrome (PWS)
Norditropin® is also indicated for the replacement of endogenous GH in adults with growth hormone deficiency (GHD)
Important Safety Information for Norditropin®
Contraindications
Norditropin® is contraindicated in patients with:
- Acute critical illness after open heart surgery, abdominal surgery or multiple accidental trauma, or those with acute respiratory failure due to the risk of increased mortality with use of pharmacologic doses of somatropin
- Pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to the risk of sudden death
- Active Malignancy
- Hypersensitivity to Norditropin® or any of its excipients. Systemic hypersensitivity reactions have been reported with postmarketing use of somatropins
- Active proliferative or severe non-proliferative diabetic retinopathy
- Pediatric patients with closed epiphyses
Warnings and Precautions
- Increased mortality in patients with acute critical illness due to complications following open heart or abdominal surgery or multiple accidental trauma, or those with respiratory failure has been reported.
- Sudden death in pediatric patients with Prader-Willi Syndrome has been reported after initiating treatment with somatropin with one or more of the following risk factors: severe obesity, history of upper airway obstruction or sleep apnea, or unidentified respiratory infection. Evaluate patients for signs of upper airway obstruction and sleep apnea before initiation of treatment.
- Increased risk of neoplasms: Monitor patients with preexisting tumors for progression or recurrence. In childhood cancer survivors who were treated with radiation to the brain/head for their first neoplasm and who developed subsequent GHD and were treated with somatropin, an increased risk of a second neoplasm, in particular meningiomas, has been reported. Pediatric patients with certain rare genetic causes of short stature have an increased risk of developing malignancies and should be carefully monitored for development of neoplasms. Monitor patients carefully for increased growth, or potential malignant changes of preexisting nevi.
- Glucose intolerance and diabetes mellitus: Treatment with somatropin may decrease insulin sensitivity, particularly at higher doses. New-onset type 2 diabetes mellitus has been reported. Monitor glucose levels in all patients. Doses of concurrent antidiabetic drugs may require adjustment.
- Intracranial hypertension has been reported in a small number of patients, usually within the first 8 weeks of somatropin treatment. Funduscopic examination should be performed before initiating treatment and periodically thereafter.
- Severe hypersensitivity: Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported with postmarketing use of somatropins.
- Fluid retention in adults (clinically manifesting as edema, arthralgia, myalgia, nerve compression syndromes including carpal tunnel syndrome/paraesthesias) may frequently occur and is usually transient and dose dependent.
- Hypoadrenalism: Patients who have or are at risk for pituitary hormone deficiency(s) may be at risk for reduced serum cortisol levels and/or unmasking of central (secondary) hypoadrenalism. In addition, patients treated with glucocorticoid replacement for previously diagnosed hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Norditropin® treatment.
- Hypothyroidism if undiagnosed/untreated, may prevent an optimal response to Norditropin®, in particular, the growth response in pediatric patients. In patients with GHD, central (secondary) hypothyroidism may first become evident or worsen during somatropin treatment. Periodic thyroid function tests and thyroid hormone replacement therapy should be initiated or adjusted when indicated.
- Slipped capital femoral epiphysis in pediatric patients may occur more frequently in patients with endocrine disorders or in patients undergoing rapid growth. Slipped capital femoral epiphysis may lead to osteonecrosis. Cases of slipped capital femoral epiphysis with or without osteonecrosis have been reported in pediatric patients with short stature receiving somatropin, including Norditropin®. Evaluate pediatric patients receiving Norditropin® with the onset of a limp or complaints of hip or knee pain for slipped capital femoral epiphysis and osteonecrosis and manage accordingly.
- Progression of preexisting scoliosis in pediatric patients can occur in patients who experience rapid growth. Patients with a history of scoliosis should be monitored for progression.
- Pancreatitis: Cases of pancreatitis have been reported. Pancreatitis should be considered in any patient who develops persistent severe abdominal pain.
- Lipoatrophy: Tissue atrophy may result when somatropin is administered subcutaneously at the same site over a long period of time. Rotate injection sites when administering Norditropin® to reduce this risk.
Adverse Reactions
- Common adverse reactions in adults and pediatric patients include: upper respiratory infection, fever, pharyngitis, headache, otitis media, edema, arthralgia, paresthesia, myalgia, peripheral edema, flu syndrome, and impaired glucose tolerance
Drug Interactions
- Glucocorticoids: Patients treated with glucocorticoid for hypoadrenalism may require an increase in their maintenance or stress doses following initiation of Norditropin®
- Pharmacologic Glucocorticoid Therapy and Supraphysiologic Glucocorticoid Treatment: Adjust glucocorticoid replacement dosing in pediatric patients receiving glucocorticoid treatment to avoid both hypoadrenalism and an inhibitory effect on growth
- Cytochrome P450-Metabolized Drugs: Norditropin® may alter the clearance. Monitor carefully if used with Norditropin®
- Oral Estrogen: Larger doses of Norditropin® may be required
- Insulin and/or Other Hypoglycemic Agents: Dose adjustment of insulin or hypoglycemic agent may be required
Use in Specific Populations
- Pregnancy and Nursing Mothers: There are limited data with somatropin use in pregnant women and nursing mothers to inform a drug-associated risk for adverse developmental outcomes
- Geriatric Use: The safety and effectiveness in patients aged 65 and over has not been evaluated in clinical studies
Please click here for Norditropin® Prescribing Information.
References:
- Data on File. Novo Nordisk, Inc.; Plainsboro, NJ.
- Norditropin [prescribing information]. Plainsboro, NJ: Novo Nordisk Inc.