Cardiovascular outcomes trial for Victoza®
LEADER—a landmark CVOT for Victoza®1
9340
patients
Key inclusion criteria:
T2D, A1C ≥7.0%
Age ≥50 years and established CVD
OR
Age ≥60 years and risk factors for CVD

Cardiovascular standards of care
(antihypertensives, lipid-lowering agents, and antiplatelet therapy)
Diabetes standards of care
(lifestyle modification, OADs, and insulin)
Duration 3.5-5 years
Time to first major adverse cardiovascular event (MACE) composed of:
Composite primary endpoint
CV death
Nonfatal MI
Nonfatal stroke

- Prospectively designed and powered to assess noninferiority and then superiority
- Patients were titrated to maximum tolerated dose of 0.6 mg to 1.8 mg
- Median daily dose of Victoza® was 1.78 mg
Victoza® significantly reduced MACE in adults with T2D and established CVD1

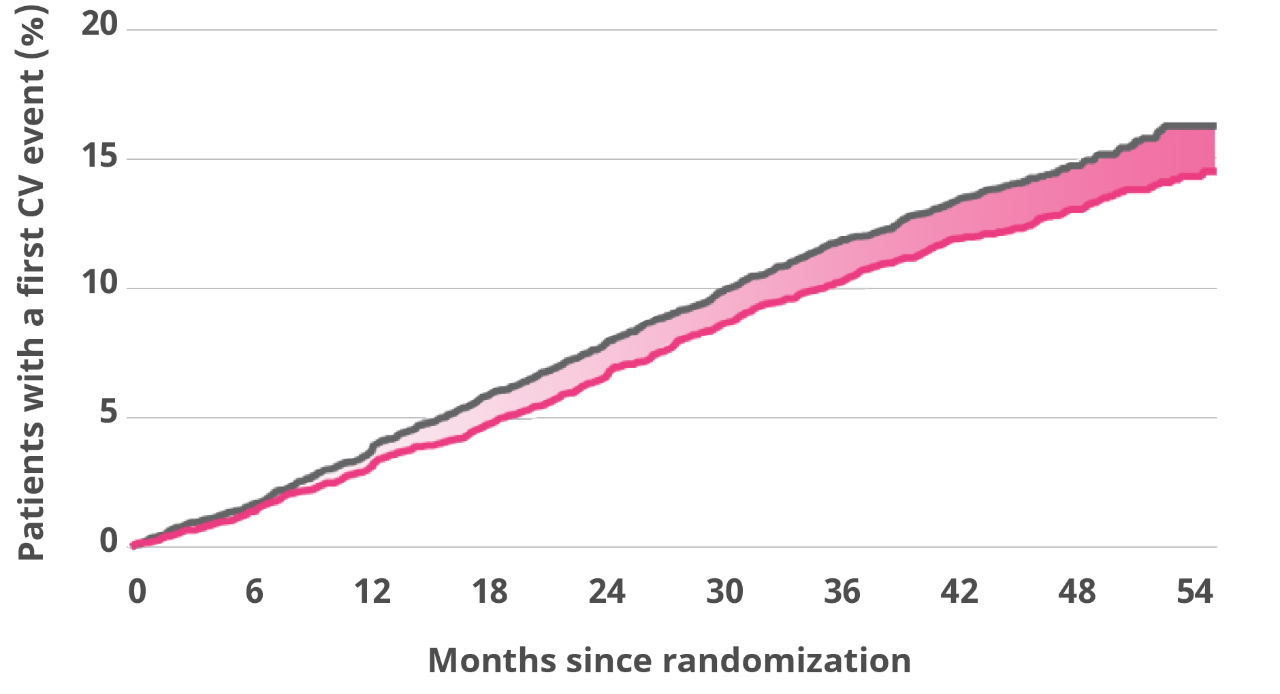

Absolute risk reduction:
1.9%

Median time of exposure to treatment: 3.5 years.
Median daily dose of Victoza®: 1.78 mg.
The primary composite outcome occurred in fewer patients in the Victoza® group (608 of 4668 patients [13.0%]) than in the SOC group (694 of 4672 [14.9%]) (hazard ratio, 0.87; 95% confidence interval [CI], 0.78-0.97; P=0.01).
Results for all 3 components of the primary composite endpoint supported the use of Victoza® vs SOC alone
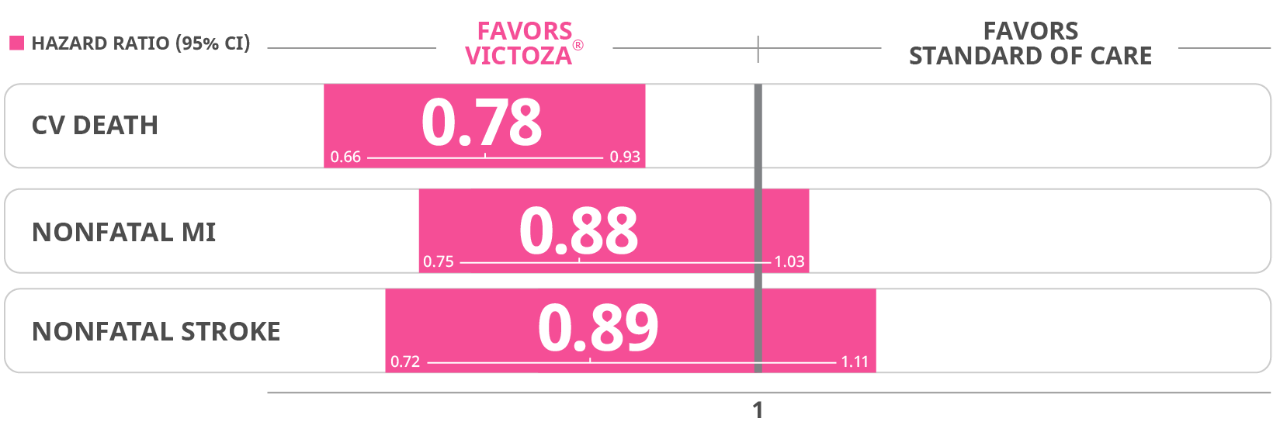
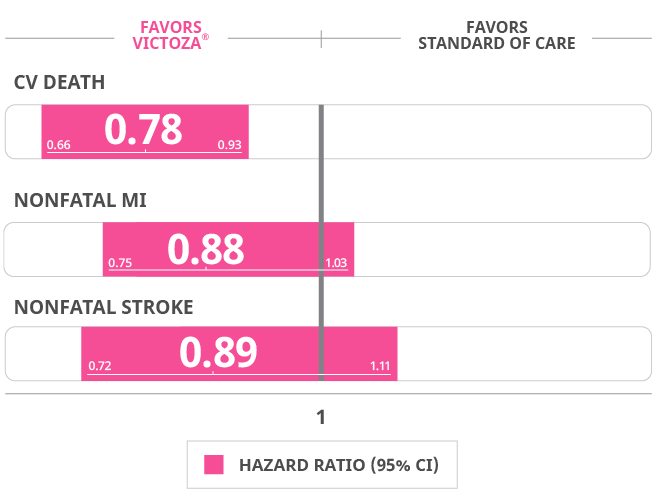
Cardiovascular death: Victoza 4.7% (219 events) Placebo 6% (278 events)
Non-fatal myocardial infarction: Victoza 6.0% (281 events) placebo 6.8% (317 events)
Non-fatal stroke: Victoza 3.4% (159 events) Placebo 3.8% (177 events)
In the LEADER trial
Victoza® provided a life-saving benefit1
Secondary endpoint: Death from CV causes in adults with T2D and established CVD

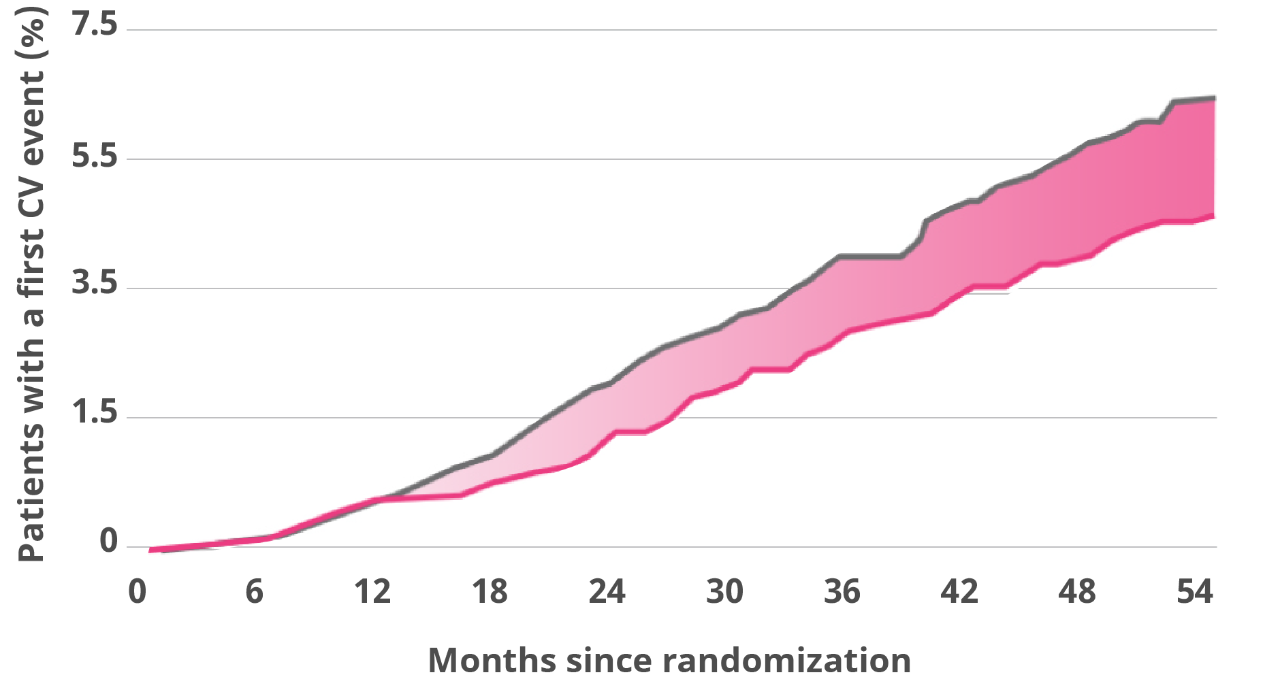
Death from CV causes was a prespecified secondary endpoint.
Hazard ratio, 0.78 (95% CI, 0.66-0.93).
Median time of exposure to treatment: 3.5 years.
Median daily dose of Victoza®: 1.78 mg.

Absolute risk reduction:
1.3%
Death from CV causes occurred in fewer patients in the Victoza® group (219 patients [4.7%]) than in the SOC group (278 [6.0%]) (hazard ratio, 0.78; 95% CI, 0.66-0.93).


Absolute risk reduction:
1.4%
Death from all causes occurred in fewer patients in the Victoza® group (381 patients [8.2%]) than in the SOC group (447 patients [9.6%]) (hazard ratio, 0.85; 95% CI, 0.74-0.97).
CV=cardiovascular; CVOT=cardiovascular outcomes trial; MI=myocardial infarction; RRR=relative risk reduction; SOC=standard of care; T2D=type 2 diabetes.
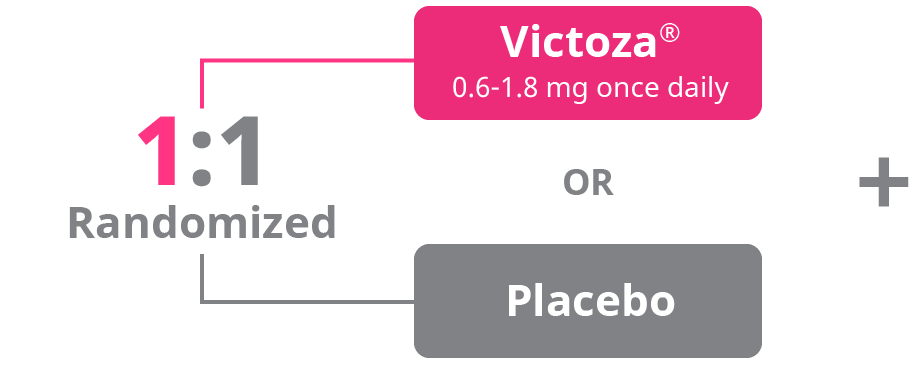
LEADER study design1
A phase 3b, multicenter, international, randomized, double-blind, placebo-controlled study to assess the cardiovascular safety of Victoza®. Adult patients with type 2 diabetes at high risk for cardiovascular events (N=9340) were randomized to receive once-daily Victoza® (0.6 mg-1.8 mg once daily with all patients being titrated to 1.8 mg once daily) or placebo. The primary endpoint was the time from randomization to a composite outcome consisting of the first occurrence of cardiovascular death, nonfatal myocardial infarction, or nonfatal stroke. The trial was prospectively designed and powered to assess whether Victoza® was noninferior or superior to placebo when measuring effect on CV events. Patients received trial products in addition to standard of care treatments such as oral antidiabetic treatments, insulin, and antihypertensive, antiplatelet, and lipid-lowering therapies.
Questions about dosing?
Learn more about prescribing Victoza®, and help your patients improve glycemic control.
Understand Victoza® efficacy and safety
When choosing a treatment for your patients with T2D, review the Victoza® study data.
Important Safety Information for Victoza®
WARNING: RISK OF THYROID C-CELL TUMORS
- Liraglutide causes dose-dependent and treatment-duration-dependent thyroid C-cell tumors at clinically relevant exposures in both genders of rats and mice. It is unknown whether Victoza® causes thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans, as the human relevance of liraglutide-induced rodent thyroid C-cell tumors has not been determined
- Victoza® is contraindicated in patients with a personal or family history of MTC and in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Counsel patients regarding the potential risk for MTC with the use of Victoza® and inform them of symptoms of thyroid tumors (eg, a mass in the neck, dysphagia, dyspnea, persistent hoarseness). Routine monitoring of serum calcitonin or using thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with Victoza®
Indications and Limitations of Use
Victoza® (liraglutide) injection 1.2 mg or 1.8 mg is indicated as an adjunct to diet and exercise to improve glycemic control in adults and pediatric patients aged 10 years and older with type 2 diabetes and to reduce the risk of major adverse cardiovascular events (cardiovascular death, non-fatal myocardial infarction, or non-fatal stroke) in adults with type 2 diabetes and established cardiovascular disease.
- Victoza® should not be used in patients with type 1 diabetes
- Victoza® contains liraglutide and should not be coadministered with other liraglutide-containing products
Important Safety Information cont.
Contraindications
- Victoza® is contraindicated in patients with a personal or family history of MTC or in patients with MEN 2, and in patients with a serious hypersensitivity reaction to liraglutide or to any of the excipients in Victoza®. Serious hypersensitivity reactions including anaphylactic reactions and angioedema have been reported with Victoza®
Warnings and Precautions
- Risk of Thyroid C-cell Tumors: If serum calcitonin is measured and found to be elevated or thyroid nodules are noted on physical examination or neck imaging, the patient should be further evaluated
- Pancreatitis: Acute pancreatitis, including fatal and nonfatal hemorrhagic or necrotizing pancreatitis, has been observed in patients treated with Victoza® postmarketing. Observe patients carefully for signs and symptoms of pancreatitis (persistent severe abdominal pain, sometimes radiating to the back with or without vomiting). If pancreatitis is suspected, Victoza® should promptly be discontinued and appropriate management should be initiated. If pancreatitis is confirmed, Victoza® should not be restarted
- Never Share a Victoza® Pen Between Patients, even if the needle is changed. Pen-sharing poses a risk for transmission of blood-borne pathogens
- Hypoglycemia: Adult patients receiving Victoza® in combination with an insulin secretagogue (eg, sulfonylurea) or insulin may have an increased risk of hypoglycemia, including severe hypoglycemia. In pediatric patients 10 years of age and older, the risk of hypoglycemia was higher with Victoza® regardless of insulin and/or metformin use
- Acute Kidney Injury: Acute renal failure and worsening of chronic renal failure, which may sometimes require hemodialysis, have been reported postmarketing, usually in association with nausea, vomiting, diarrhea, or dehydration. Use caution when initiating or escalating doses of Victoza® in patients with renal impairment
- Hypersensitivity Reactions: There have been postmarketing reports of serious hypersensitivity reactions (e.g., anaphylactic reactions and angioedema) in patients treated with Victoza®. If a hypersensitivity reaction occurs, discontinue Victoza®; treat promptly per standard of care, and monitor until signs and symptoms resolve. Do not use in patients with a previous hypersensitivity reaction to Victoza® Anaphylaxis and angioedema have been reported with other GLP-1 receptor agonists. Use caution in a patient with a history of anaphylaxis or angioedema with another GLP-1 receptor agonist because it is unknown whether such patients will be predisposed to these reactions with Victoza®
- Acute Gallbladder Disease: Acute events of gallbladder disease such as cholelithiasis or cholecystitis have been reported in GLP-1 receptor agonist trials and postmarketing. In the LEADER trial, 3.1% of Victoza®-treated patients versus 1.9% of placebo-treated patients reported an acute event of gallbladder disease, such as cholelithiasis or cholecystitis. If cholelithiasis is suspected, gallbladder studies and appropriate clinical follow-up are indicated
Adverse Reactions
- The most common adverse reactions (incidence ≥5%) in clinical trials are nausea, diarrhea, vomiting, decreased appetite, dyspepsia, and constipation
- Immunogenicity-related events, including urticaria, were more common among Victoza®-treated patients (0.8%) than among comparator-treated patients (0.4%) in clinical trials
Drug Interactions
- Victoza® delays gastric emptying and may impact the absorption of concomitantly administered oral medications. Caution should be exercised when oral medications are concomitantly administered with Victoza®
- When initiating Victoza®, consider reducing the dose of concomitantly administered insulin secretagogues (such as sulfonylureas) or insulin to reduce the risk of hypoglycemia
Use in Specific Populations
- The safety and effectiveness of Victoza® have not been established in pediatric patients less than 10 years of age
- Victoza® slows gastric emptying. Victoza® has not been studied in patients with pre-existing gastroparesis
- Victoza® should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus
Please click here for Victoza® Prescribing Information, including Boxed Warning.
Reference:
- Victoza [package insert]. Plainsboro, NJ: Novo Nordisk Inc.